Atypical changes in Candida albicans cells treated with the Venetin-1 complex from earthworm coelomic fluid
- PMID: 36807384
- PMCID: PMC9938250
- DOI: 10.1038/s41598-023-29728-0
Atypical changes in Candida albicans cells treated with the Venetin-1 complex from earthworm coelomic fluid
Abstract
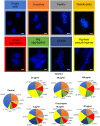
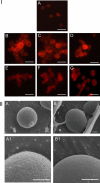
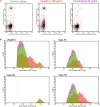
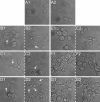
In the present research, the effect of a protein-polysaccharide complex Venetin-1 obtained from the coelomic fluid of Dendrobaena veneta earthworm on Candida albicans cells was characterized. The compound destroyed fungal cells without showing cytotoxicity to human skin fibroblasts, which was demonstrated in earlier studies. Since it had an effect on the fungal cell wall and membrane, this complex was compared with the known antifungal antibiotic fluconazole. Both preparations disturbed the division of yeast cells and resulted in the formation of aggregates and chains of unseparated cells, which was illustrated by staining with fluorochromes. Fluorescent staining of the cell wall with Calcofluor white facilitated comparison of the types of aggregates formed after the action of both substances. The analysis performed with the use of Congo red showed that Venetin-1 exposed deeper layers of the cell wall, whereas no such effect was visible after the use of fluconazole. The FTIR analysis confirmed changes in the mannoprotein layer of the cell wall after the application of the Venetin-1 complex. Staining with Rhodamine 123 and the use of flow cytometry allowed comparison of changes in the mitochondria. Significantly elongated mitochondria were observed after the Venetin-1 application, but not after the application of the classic antibiotic. Phase contrast microscopy revealed vacuole enlargement after the Venetin-1 application. The flow cytometry analysis of C. albicans cells treated with Venetin-1 and fluconazole showed that both substances caused a significant decrease in cell viability.
© 2023. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures





Similar articles
-
Autophagy of Candida albicans cells after the action of earthworm Venetin-1 nanoparticle with protease inhibitor activity.Sci Rep. 2023 Aug 30;13(1):14228. doi: 10.1038/s41598-023-41281-4. Sci Rep. 2023. PMID: 37648723 Free PMC article.
-
Anti-Candida albicans effect of the protein-carbohydrate fraction obtained from the coelomic fluid of earthworm Dendrobaena veneta.PLoS One. 2019 Mar 11;14(3):e0212869. doi: 10.1371/journal.pone.0212869. eCollection 2019. PLoS One. 2019. PMID: 30856188 Free PMC article.
-
Candida albicans cell wall as a target of action for the protein-carbohydrate fraction from coelomic fluid of Dendrobaena veneta.Sci Rep. 2020 Oct 1;10(1):16352. doi: 10.1038/s41598-020-73044-w. Sci Rep. 2020. PMID: 33004852 Free PMC article.
-
Candida and candidaemia. Susceptibility and epidemiology.Dan Med J. 2013 Nov;60(11):B4698. Dan Med J. 2013. PMID: 24192246 Review.
-
Molecular epidemiology, antifungal susceptibility, and ERG11 gene mutation of Candida species isolated from vulvovaginal candidiasis: Comparison between recurrent and non-recurrent infections.Microb Pathog. 2022 Sep;170:105696. doi: 10.1016/j.micpath.2022.105696. Epub 2022 Jul 31. Microb Pathog. 2022. PMID: 35921954 Review.
Cited by
-
Morphological and environmental analysis of the glacier ice alga Ancylonema alaskanum.Sci Rep. 2025 May 27;15(1):18578. doi: 10.1038/s41598-025-95754-9. Sci Rep. 2025. PMID: 40425824 Free PMC article.
-
Autophagy of Candida albicans cells after the action of earthworm Venetin-1 nanoparticle with protease inhibitor activity.Sci Rep. 2023 Aug 30;13(1):14228. doi: 10.1038/s41598-023-41281-4. Sci Rep. 2023. PMID: 37648723 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

