Cost-effectiveness of human T-cell leukemia virus type 1 (HTLV-1) antenatal screening for prevention of mother-to-child transmission
- PMID: 36809372
- PMCID: PMC9983854
- DOI: 10.1371/journal.pntd.0011129
Cost-effectiveness of human T-cell leukemia virus type 1 (HTLV-1) antenatal screening for prevention of mother-to-child transmission
Abstract
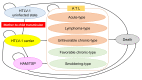
Background: Human T-cell leukemia virus type 1 (HTLV-1) causes adult T-cell leukemia-lymphoma (ATL) and HTLV-1-associated myelopathy-tropical spastic paraparesis (HAM/TSP) with a poor prognosis. This study aimed to evaluate the cost-effectiveness and health impact of HTLV-1 antenatal screening.
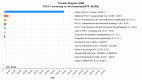
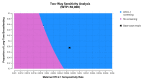
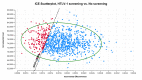
Methodology/principal findings: A state-transition model was developed for HTLV-1 antenatal screening and no screening over a lifetime horizon from a healthcare payer perspective. A hypothetical cohort of 30-year-old individuals was targeted. The main outcomes were costs, quality-adjusted life-years (QALYs), life expectancy life-years (LYs), incremental cost-effectiveness ratios (ICERs), HTLV-1 carriers, ATL cases, HAM/TSP cases, ATL-associated deaths, and HAM/TSP-associated deaths. The willingness-to-pay (WTP) threshold was set at US$50,000 per QALY gained. In the base-case analysis, HTLV-1 antenatal screening (US$76.85, 24.94766 QALYs, 24.94813 LYs, ICER; US$40,100 per QALY gained) was cost-effective compared with no screening (US$2.18, 24.94580 QALYs, 24.94807 LYs). Cost-effectiveness was sensitive to the maternal HTLV-1 seropositivity rate, HTLV-1 transmission rate with long-term breastfeeding from HTLV-1 seropositive mothers to children, and the cost of the HTLV-1 antibody test. HTLV-1 antenatal screening was cost-effective when the maternal HTLV-1 seropositivity rate was greater than 0.0022 and the cost of the HTLV-1 antibody test was lower than US$94.8. Probabilistic sensitivity analysis using a second-order Monte-Carlo simulation showed that HTLV-1 antenatal screening was 81.1% cost-effective at a WTP threshold of US$50,000 per QALY gained. For 10,517,942 individuals born between 2011 and 2021, HTLV-1 antenatal screening costs US$785 million, increases19,586 QALYs and 631 LYs, and prevents 125,421 HTLV-1 carriers, 4,405 ATL cases, 3,035 ATL-associated deaths, 67 HAM/TSP cases, and 60 HAM/TSP-associated deaths, compared with no screening over a lifetime.
Conclusion/significance: HTLV-1 antenatal screening is cost-effective and has the potential to reduce ATL and HAM/TSP morbidity and mortality in Japan. The findings strongly support the recommendation for HTLV-1 antenatal screening as a national infection control policy in HTLV-1 high-prevalence countries.
Copyright: © 2023 Akiko Kowada. This is an open access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Conflict of interest statement
The author has declared that no competing interests exist.
Figures

References
-
- Nagasaka M, Yamagishi M, Yagishita N, Araya N, Kobayashi S, Makiyama J, et al. Mortality and risk of progression to adult T cell leukemia/lymphoma in HTLV-1-associated myelopathy/tropical spastic paraparesis. Proc Natl Acad Sci. U S A. 2020;117:11685–11691. doi: 10.1073/pnas.1920346117 - DOI - PMC - PubMed
MeSH terms
LinkOut - more resources
Full Text Sources
Miscellaneous

