Impact of Type 2 Diabetes Duration on the Efficacy and Safety of Add-on Lixisenatide in Asian Individuals Receiving Basal Insulin: A Pooled Analysis
- PMID: 36809495
- PMCID: PMC10064411
- DOI: 10.1007/s13300-023-01369-6
Impact of Type 2 Diabetes Duration on the Efficacy and Safety of Add-on Lixisenatide in Asian Individuals Receiving Basal Insulin: A Pooled Analysis
Abstract
Introduction: This analysis investigated the efficacy and safety of add-on lixisenatide by disease duration in Asian people with type 2 diabetes inadequately controlled with basal insulin ± oral antidiabetic drugs.
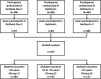
Methods: Data for Asian participants in the GetGoal-Duo 1, GetGoal-L, and GetGoal-L-C studies were pooled and categorized by diabetes duration: < 10 years (group 1), 10 to < 15 years (group 2), and ≥ 15 years (group 3). Efficacy and safety of lixisenatide versus placebo were evaluated by subgroup. The potential influence of diabetes duration on efficacy was examined using multivariable regression analyses.
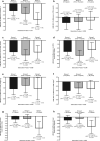
Results: A total of 555 participants were included (mean age 53.9 years, 52.4% male). No significant differences in treatment effect between the duration subgroups were observed for the changes from baseline to 24 weeks in glycated hemoglobin (HbA1c), fasting plasma glucose (FPG), postprandial glucose (PPG), PPG excursion, body weight or body mass index, or the proportion of participants with HbA1c < 7% at 24 weeks (all P values for interaction > 0.1). Change in insulin dosage (U/day) was significantly different between subgroups (P = 0.038). Multivariable regression analysis showed participants in group 1 had a smaller change in body weight and basal insulin dose over the 24-week treatment period than participants in group 3 (P = 0.014 and 0.030, respectively) and were less likely to achieve an HbA1c < 7% than participants in group 2 (P = 0.047). No severe hypoglycemia was reported. A higher proportion of participants in group 3 versus the other groups had symptomatic hypoglycemia, for both lixisenatide and placebo, and T2D duration had a significant effect on hypoglycemia risk (P = 0.001).
Conclusions: Lixisenatide improved glycemic control in Asian individuals regardless of diabetes duration, without increasing the risk of hypoglycemia. Individuals with longer disease duration had a greater risk of symptomatic hypoglycemia than individuals with shorter disease duration regardless of treatment. No additional safety concerns were observed.
Clinical trial registration: GetGoal-Duo 1, ClinicalTrials.gov record NCT00975286; GetGoal-L, ClinicalTrials.gov record NCT00715624; GetGoal-L-C, ClinicalTrials.gov record NCT01632163.
Keywords: Disease duration; Glucagon-like peptide 1 receptor agonist; Lixisenatide; Pooled analysis; Type 2 diabetes.
© 2023. The Author(s).
Figures
References
-
- Moghissi ES. Treating patients with diabetes of long duration: GLP-1 receptor agonists and insulin in combination. J Am Osteopath Assoc. 2014;114(5 Suppl 2):S22–S29. - PubMed
-
- Turner RC, Cull CA, Frighi V, Holman RR, UK Prospective Diabetes Study (UKPDS) Glycemic control with diet, sulfonylurea, metformin, or insulin in patients with type 2 diabetes mellitus: progressive requirement for multiple therapies (UKPDS 49) JAMA. 1999;281(21):2005–2012. doi: 10.1001/jama.281.21.2005. - DOI - PubMed
-
- Buse JB, Wexler DJ, Tsapas A, et al. 2019 update to: management of hyperglycemia in type 2 diabetes, 2018. A Consensus Report by the American Diabetes Association (ADA) and the European Association for the Study of Diabetes (EASD) Diabetes Care. 2020;43(2):487–493. doi: 10.2337/dci19-0066. - DOI - PMC - PubMed
Associated data
LinkOut - more resources
Full Text Sources
Medical

