L-carnitine attenuated hyperuricemia-associated left ventricular remodeling through ameliorating cardiomyocytic lipid deposition
- PMID: 36817129
- PMCID: PMC9929955
- DOI: 10.3389/fphar.2023.1016633
L-carnitine attenuated hyperuricemia-associated left ventricular remodeling through ameliorating cardiomyocytic lipid deposition
Abstract
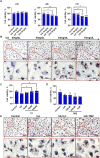
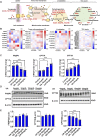
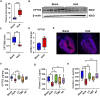
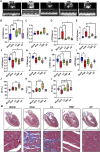
Hyperuricemia (HUA) is associated with left ventricular remodeling (LVR) and thereby causes the initiation and development of a large number of cardiovascular diseases. LVR is typically accompanied by cardiomyocyte energy metabolic disorder. The energy supply of cardiomyocytes is provided by glucose and fatty acid (FA) metabolism. Currently, the effect of HUA on cardiomyocytic FA metabolism is unclear. In this study, we demonstrate that UA-induced cardiomyocyte injury is associated with cytoplasmic lipid deposition, which can be ameliorated by the FA metabolism-promoting drug L-carnitine (LC). UA suppresses carnitine palmitoyl transferase 1B (CPT1B), thereby inhibiting FA transport into the mitochondrial inner matrix for elimination. LC intervention can ameliorate HUA-associated left ventricular anterior wall thickening in mice. This study showed that FA transport dysfunction plays is a critical mechanism in both cardiomyocytic injury and HUA-associated LVR and promoting cytoplasmic FA transportation through pharmacological treatment by LC is a valid strategy to attenuate HUA-associated LVR.
Keywords: L-carnitine; cardiomyocytes; fatty acid; hyperuricemia; ventricular remodeling.
Copyright © 2023 Yang, Lin, Zheng, Zhang, Li, Huang, Wu, Zhao, Li, Luo, Jiang, Zhang, Wang, Xia and Pang.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures


References
LinkOut - more resources
Full Text Sources

