Precision-engineering of subunit vaccine particles for prevention of infectious diseases
- PMID: 36817419
- PMCID: PMC9935699
- DOI: 10.3389/fimmu.2023.1131057
Precision-engineering of subunit vaccine particles for prevention of infectious diseases
Abstract
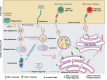
Vaccines remain the best approach for the prevention of infectious diseases. Protein subunit vaccines are safe compared to live-attenuated whole cell vaccines but often show reduced immunogenicity. Subunit vaccines in particulate format show improved vaccine efficacy by inducing strong immune responses leading to protective immunity against the respective pathogens. Antigens with proper conformation and function are often required to induce functional immune responses. Production of such antigens requiring post-translational modifications and/or composed of multiple complex domains in bacterial hosts remains challenging. Here, we discuss strategies to overcome these limitations toward the development of particulate vaccines eliciting desired humoral and cellular immune responses. We also describe innovative concepts of assembling particulate vaccine candidates with complex antigens bearing multiple post-translational modifications. The approaches include non-covalent attachments (e.g. biotin-avidin affinity) and covalent attachments (e.g. SpyCatcher-SpyTag) to attach post-translationally modified antigens to particles.
Keywords: cross-presentation; dendritic cells; infectious diseases; particulate vaccine; post-translational modification; protective immunity.
Copyright © 2023 Chen, Pounraj, Sivakumaran, Kakkanat, Sam, Kabir and Rehm.
Conflict of interest statement
Author BHAR is an inventor of the biopolymer particle technology and co-founder/shareholder of Polybatics Ltd. BHAR and SC are inventors on a patent application describing the immunogenic carrier protein CRM197 vaccine platform and its uses. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures
References
-
- Seyfoori A, Shokrollahi Barough M, Mokarram P, Ahmadi M, Mehrbod P, Sheidary A, et al. . Emerging advances of nanotechnology in drug and vaccine delivery against viral associated respiratory infectious diseases (Varid). Int J Mol Sci (2021) 22(13):6937. doi: 10.3390/ijms22136937 - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical