Recent development in multizonal scaffolds for osteochondral regeneration
- PMID: 36817819
- PMCID: PMC9931622
- DOI: 10.1016/j.bioactmat.2023.01.012
Recent development in multizonal scaffolds for osteochondral regeneration
Abstract
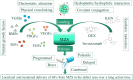
Osteochondral (OC) repair is an extremely challenging topic due to the complex biphasic structure and poor intrinsic regenerative capability of natural osteochondral tissue. In contrast to the current surgical approaches which yield only short-term relief of symptoms, tissue engineering strategy has been shown more promising outcomes in treating OC defects since its emergence in the 1990s. In particular, the use of multizonal scaffolds (MZSs) that mimic the gradient transitions, from cartilage surface to the subchondral bone with either continuous or discontinuous compositions, structures, and properties of natural OC tissue, has been gaining momentum in recent years. Scrutinizing the latest developments in the field, this review offers a comprehensive summary of recent advances, current hurdles, and future perspectives of OC repair, particularly the use of MZSs including bilayered, trilayered, multilayered, and gradient scaffolds, by bringing together onerous demands of architecture designs, material selections, manufacturing techniques as well as the choices of growth factors and cells, each of which possesses its unique challenges and opportunities.
Keywords: Cells; Fabrication; Growth factors; Multizonal scaffolds; Osteochondral regeneration.
© 2023 The Authors.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures








References
-
- Murray C.J.L., Lopez A.D., World Health O., World B., Harvard H. In: In 1990 and Projected to 2020 : Summary/ Murray Christopher J.L., Lopez Alan D., editors. World Health Organization; Geneva: 1996. School of Public, the Global burden of disease : a comprehensive assessment of mortality and disability from diseases, injuries, and risk factors.
-
- eumusc.net, Musculoskeletal Health in Europe, Report v5.0. - PubMed
-
- Baumann C., Hinckel B., Bozynski C., Farr J. A Clinical Casebook; 2019. Articular Cartilage: Structure and Restoration; pp. 3–24.
-
- Deng C., Xu C., Zhou Q., Cheng Y. vol. 11. Wiley Interdiscip Rev Nanomed Nanobiotechnol; 2019. (Advances of Nanotechnology in Osteochondral Regeneration). - PubMed
Publication types
LinkOut - more resources
Full Text Sources

