Sea urchin (Diadema savignyi) extract as a novel protective agent against cisplatin induced neurotoxicity in rats
- PMID: 36823675
- PMCID: PMC9948489
- DOI: 10.1186/s40360-023-00651-4
Sea urchin (Diadema savignyi) extract as a novel protective agent against cisplatin induced neurotoxicity in rats
Abstract
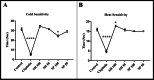
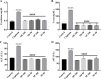
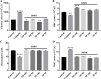
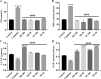
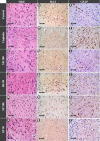
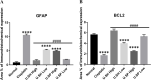
Neurotoxicity is a severe side effect of platinum compounds used for cancer chemotherapy such as Cisplatin. This neurotoxicity leads to severe cognitive and nervous dysfunction, therefore, limiting the dose of Cisplatin and compromising the treatment protocol.The present study investigates the neuroprotective effect of Sea Urchins which is a marine animal known for its rich bioactive compounds. Male Sprague Dawley rats received Cisplatin (2 mg/kg body weight) for 4 weeks, two times per week, followed by Sea Urchin extracts (50 and 100 mg/kg body weight) twice weekly for 4 weeks.Results show that rats treated with Urchin's extracts showed a significant improvement in the thermal (heat and cold) sensitivity compared to untreated rats. Liver enzymes Alanine Aminotransferase (ALT) and Aspartate Aminotransferase (AST) and Urea levels were also significantly decreased back to normal following treatment with sea urchin extracts. Brain tissue oxidative stress marker Nitric oxide (NO) and lipid peroxidation marker Malondialdehyde (MDA) increased significantly in the cisplatin-treated rats while the reduced glutathione levels (GSH) and catalase activity (CAT) showed a significant decrease. Treatment with sea Urchin extracts reversed these changes.Histological and immunohistochemical examination of the cerebral cortex reveled degenerative changes such as karyopyknosis and shrunken necrotic ghost like neurons in the cisplatin treated groups. There was also strong positive Glial fibrillary acidic protein (GFAP) reactivity and a negative B-cell leukemia/lymphoma 2 protein (Bcl2) reaction in most apparent neurons, indicating strong apoptotic changes. Treatment with Urchin extracts reversed these changes. Quantification of cerebral cortex neurons also revealed the strong effect of the extracts. Cisplatin treated groups showed 3708 cells/ mm3 compared to 8091 cells/mm3 in the normal rats. Extract treatment increased the neuronal numbers to almost normal levels. Quantification of the Immuno-histochemical expression of GFAP showed an increase by 10-folds after cisplatin administration. A remarkable decline from the cisplatin group was seen in the extract treated groups.In Conclusion, Sea Urchins extracts possess a strong neuroprotective activity and could provide a novel therapeutic method to prevent Cisplatin-induced neurotoxicity.
Keywords: Bcl2; Cisplatin; Cisplatin induced toxicity; Diadema Savignyi; GFAP; Neurotoxicity; Oxidative stress; Sea urchins.
© 2023. The Author(s).
Conflict of interest statement
All authors declare no competing interests financial or personal nature.
Figures

Similar articles
-
Neuroprotective effect of chrysin on isoniazid-induced neurotoxicity via suppression of oxidative stress, inflammation and apoptosis in rats.Neurotoxicology. 2020 Dec;81:197-208. doi: 10.1016/j.neuro.2020.10.009. Epub 2020 Oct 26. Neurotoxicology. 2020. PMID: 33121995
-
Hepatoprotective effect of curcumin and alpha-tocopherol against cisplatin-induced oxidative stress.BMC Complement Altern Med. 2014 Mar 28;14:111. doi: 10.1186/1472-6882-14-111. BMC Complement Altern Med. 2014. PMID: 24674233 Free PMC article.
-
Neuroprotective effects of carvacrol against cadmium-induced neurotoxicity in rats: role of oxidative stress, inflammation and apoptosis.Metab Brain Dis. 2022 Apr;37(4):1259-1269. doi: 10.1007/s11011-022-00945-2. Epub 2022 Mar 22. Metab Brain Dis. 2022. PMID: 35316447
-
Protective effect of Schisandra chinensis bee pollen extract on liver and kidney injury induced by cisplatin in rats.Biomed Pharmacother. 2017 Nov;95:1765-1776. doi: 10.1016/j.biopha.2017.09.083. Epub 2017 Oct 6. Biomed Pharmacother. 2017. PMID: 28962082
-
[Medical and prophylactic properties lipids and antioxidants derived from sea hydrobionts].Vopr Pitan. 2011;80(2):4-8. Vopr Pitan. 2011. PMID: 21692341 Review. Russian.
Cited by
-
Paracentrotus lividus sea urchin gonadal extract mitigates neurotoxicity and inflammatory signaling in a rat model of Parkinson's disease.PLoS One. 2024 Dec 18;19(12):e0315858. doi: 10.1371/journal.pone.0315858. eCollection 2024. PLoS One. 2024. PMID: 39693313 Free PMC article.
-
Modulation of Brain Kynurenic Acid by N-Acetylcysteine Prevents Cognitive Impairment and Muscular Weakness Induced by Cisplatin in Female Rats.Cells. 2024 Dec 2;13(23):1989. doi: 10.3390/cells13231989. Cells. 2024. PMID: 39682737 Free PMC article.
-
Paeonol protects against cisplatin-induced premature ovarian failure via anti-inflammatory and antioxidant activities: an integrated approach of network pharmacology, molecular dynamics simulation, and experimental validation.Naunyn Schmiedebergs Arch Pharmacol. 2025 Jul 24. doi: 10.1007/s00210-025-04430-2. Online ahead of print. Naunyn Schmiedebergs Arch Pharmacol. 2025. PMID: 40705082
References
-
- Scadding JW. Treatment of neuropathic pain: historical aspects. Pain Med. 2004;5 Suppl 1:S3–8. 10.1111/j.1526-4637.2004.04018.x. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

