This is a preprint.
Division of labor and cooperation between different butyrophilin proteins controls phosphoantigen-mediated activation of human γδ T cells
- PMID: 36824912
- PMCID: PMC9949253
- DOI: 10.21203/rs.3.rs-2583246/v1
Division of labor and cooperation between different butyrophilin proteins controls phosphoantigen-mediated activation of human γδ T cells
Update in
-
A distinct topology of BTN3A IgV and B30.2 domains controlled by juxtamembrane regions favors optimal human γδ T cell phosphoantigen sensing.Nat Commun. 2023 Nov 22;14(1):7617. doi: 10.1038/s41467-023-41938-8. Nat Commun. 2023. PMID: 37993425 Free PMC article.
Abstract
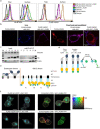
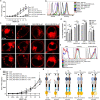
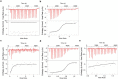
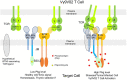
Butyrophilin (BTN)-3A and BTN2A1 molecules control TCR-mediated activation of human Vγ9Vδ2 T-cells triggered by phosphoantigens (PAg) from microbes and tumors, but the molecular rules governing antigen sensing are unknown. Here we establish three mechanistic principles of PAg-action. Firstly, in humans, following PAg binding to the BTN3A1-B30.2 domain, Vγ9Vδ2 TCR triggering involves the V-domain of BTN3A2/BTN3A3. Moreover, PAg/B30.2 interaction, and the critical γδ-T-cell-activating V-domain, localize to different molecules. Secondly, this distinct topology as well as intracellular trafficking and conformation of BTN3A heteromers or ancestral-like BTN3A homomers are controlled by molecular interactions of the BTN3 juxtamembrane region. Finally, the ability of PAg not simply to bind BTN3A-B30.2, but to promote its subsequent interaction with the BTN2A1-B30.2 domain, is essential for T-cell activation. Defining these determinants of cooperation and division of labor in BTN proteins deepens understanding of PAg sensing and elucidates a mode of action potentially applicable to other BTN/BTNL family members.
Keywords: BTN2A1; BTN3A1; Butyrophilin; TCR; Vγ9Vδ2 T cells; juxtamembrane; phosphoantigens; γδ T cells.
Conflict of interest statement
Competing interests: The authors declare no competing interests.
Figures


References
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources

