Bioinformatics analysis and consistency verification of a novel tuberculosis vaccine candidate HP13138PB
- PMID: 36825009
- PMCID: PMC9942524
- DOI: 10.3389/fimmu.2023.1102578
Bioinformatics analysis and consistency verification of a novel tuberculosis vaccine candidate HP13138PB
Erratum in
-
Corrigendum: Bioinformatics analysis and consistency verification of a novel tuberculosis vaccine candidate HP13138PB.Front Immunol. 2023 Feb 8;14:1154693. doi: 10.3389/fimmu.2023.1154693. eCollection 2023. Front Immunol. 2023. PMID: 36845146 Free PMC article.
Abstract
Background: With the increasing incidence of tuberculosis (TB) and the shortcomings of existing TB vaccines to prevent TB in adults, new TB vaccines need to be developed to address the complex TB epidemic.
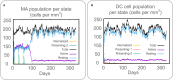
Method: The dominant epitopes were screened from antigens to construct a novel epitope vaccine termed HP13138PB. The immune properties, structure, and function of HP13138PB were predicted and analyzed with bioinformatics and immunoinformatics. Then, the immune responses induced by the HP13138PB were confirmed by enzyme-linked immunospot assay (ELISPOT) and Th1/Th2/Th17 multi-cytokine detection kit.
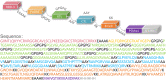
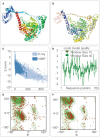
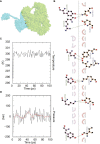
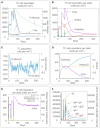
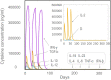
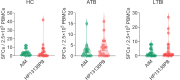
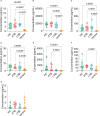
Result: The HP13138PB vaccine consisted of 13 helper T lymphocytes (HTL) epitopes, 13 cytotoxic T lymphocytes (CTL) epitopes, and 8 B-cell epitopes. It was found that the antigenicity, immunogenicity, and solubility index of the HP13138PB vaccine were 0.87, 2.79, and 0.55, respectively. The secondary structure prediction indicated that the HP13138PB vaccine had 31% of α-helix, 11% of β-strand, and 56% of coil. The tertiary structure analysis suggested that the Z-score and the Favored region of the HP13138PB vaccine were -4.47 88.22%, respectively. Furthermore, the binding energies of the HP13138PB to toll-like receptor 2 (TLR2) was -1224.7 kcal/mol. The immunoinformatics and real-world experiments showed that the HP13138PB vaccine could induce an innate and adaptive immune response characterized by significantly higher levels of cytokines such as interferon-gamma (IFN-γ), tumor necrosis factor-α (TNF-α), interleukin-4 (IL-4), and IL-10.
Conclusion: The HP13138PB is a potential vaccine candidate to prevent TB, and this study preliminarily evaluated the ability of the HP13138PB to generate an immune response, providing a precursor target for developing TB vaccines.
Keywords: bioinformatics; epitope vaccines; immune responses; immunoinformatics; tuberculosis.
Copyright © 2023 Cheng, Jiang, Wang, Wang, Xue, Wang and Gong.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures



Similar articles
-
PP19128R, a Multiepitope Vaccine Designed to Prevent Latent Tuberculosis Infection, Induced Immune Responses In Silico and In Vitro Assays.Vaccines (Basel). 2023 Apr 17;11(4):856. doi: 10.3390/vaccines11040856. Vaccines (Basel). 2023. PMID: 37112768 Free PMC article.
-
Bioinformatics Analysis and Immunogenicity Assessment of the Novel Multi-Stage DNA Vaccine W541 Against Mycobacterium Tuberculosis.Immun Inflamm Dis. 2024 Nov;12(11):e70074. doi: 10.1002/iid3.70074. Immun Inflamm Dis. 2024. PMID: 39588938 Free PMC article.
-
Evaluation of the consistence between the results of immunoinformatics predictions and real-world animal experiments of a new tuberculosis vaccine MP3RT.Front Cell Infect Microbiol. 2022 Nov 2;12:1047306. doi: 10.3389/fcimb.2022.1047306. eCollection 2022. Front Cell Infect Microbiol. 2022. PMID: 36405961 Free PMC article.
-
Bioinformatics analysis, immunogenicity, and therapeutic efficacy evaluation of a novel multi-stage, multi-epitope DNA vaccine for tuberculosis.Int Immunopharmacol. 2025 Apr 16;152:114415. doi: 10.1016/j.intimp.2025.114415. Epub 2025 Mar 13. Int Immunopharmacol. 2025. PMID: 40086060
-
[Novel vaccines against M. tuberculosis].Kekkaku. 2006 Dec;81(12):745-51. Kekkaku. 2006. PMID: 17240920 Review. Japanese.
Cited by
-
A new candidate epitope-based vaccine against PspA PhtD of Streptococcus pneumoniae: a computational experimental approach.Front Cell Infect Microbiol. 2023 Nov 15;13:1271143. doi: 10.3389/fcimb.2023.1271143. eCollection 2023. Front Cell Infect Microbiol. 2023. PMID: 38035337 Free PMC article.
-
Knowledge mapping of disease-modifying therapy (DMT) in multiple sclerosis (MS): A bibliometrics analysis.Heliyon. 2024 May 31;10(11):e31744. doi: 10.1016/j.heliyon.2024.e31744. eCollection 2024 Jun 15. Heliyon. 2024. PMID: 38868066 Free PMC article.
-
Genome-wide expression in human whole blood for diagnosis of latent tuberculosis infection: a multicohort research.Front Microbiol. 2025 May 9;16:1584360. doi: 10.3389/fmicb.2025.1584360. eCollection 2025. Front Microbiol. 2025. PMID: 40415930 Free PMC article.
-
Antigen identification strategies and preclinical evaluation models for advancing tuberculosis vaccine development.NPJ Vaccines. 2024 Mar 9;9(1):57. doi: 10.1038/s41541-024-00834-y. NPJ Vaccines. 2024. PMID: 38461350 Free PMC article. Review.
-
In-silico development of a novel TLR2-mediating multi-epitope vaccine against Mycobacterium tuberculosis.In Silico Pharmacol. 2025 Feb 25;13(1):34. doi: 10.1007/s40203-025-00322-8. eCollection 2025. In Silico Pharmacol. 2025. PMID: 40018380
References
-
- WHO . Global tuberculosis report 2022. Geneva: World Health Organization: Genevapp: World Health Organization; (2022).
-
- Huang F, Zhao Y. Global control of tuberculosis: Current status and future prospects. In: Zoonoses (2021) 2:21. doi: 10.15212/ZOONOSES-2021-0021 - DOI
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

