NO and Heme Proteins: Cross-Talk between Heme and Cysteine Residues
- PMID: 36829880
- PMCID: PMC9952723
- DOI: 10.3390/antiox12020321
NO and Heme Proteins: Cross-Talk between Heme and Cysteine Residues
Abstract
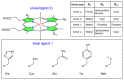
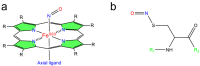
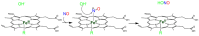
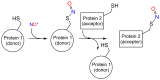
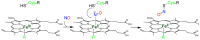
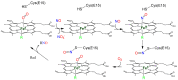
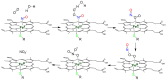
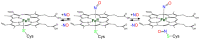
Heme proteins are a diverse group that includes several unrelated families. Their biological function is mainly associated with the reactivity of the heme group, which-among several other reactions-can bind to and react with nitric oxide (NO) and other nitrogen compounds for their production, scavenging, and transport. The S-nitrosylation of cysteine residues, which also results from the reaction with NO and other nitrogen compounds, is a post-translational modification regulating protein activity, with direct effects on a variety of signaling pathways. Heme proteins are unique in exhibiting this dual reactivity toward NO, with reported examples of cross-reactivity between the heme and cysteine residues within the same protein. In this work, we review the literature on this interplay, with particular emphasis on heme proteins in which heme-dependent nitrosylation has been reported and those for which both heme nitrosylation and S-nitrosylation have been associated with biological functions.
Keywords: S-nitrosylation; cysteine; heme; heme proteins; nitric oxide; nitrosation.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


References
Publication types
LinkOut - more resources
Full Text Sources

