Antioxidant Activity Enhancement Effect of Silver-Ionized Water: Silver Cation Prepared by Electrolysis
- PMID: 36830025
- PMCID: PMC9952159
- DOI: 10.3390/antiox12020467
Antioxidant Activity Enhancement Effect of Silver-Ionized Water: Silver Cation Prepared by Electrolysis
Abstract
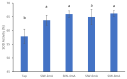
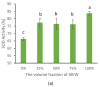
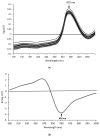
In the present study, tap water, alkaline electrolyzed water (AlEW) and tourmaline water (TMW) were used as the electrolytes to generated the silver-ionized water (SIW), AlEW-SIW and TMW-SIW, respectively. The antioxidant properties of the samples containing ascorbic acid (AsA) were investigated by WST-kit method. The results showed that the SOD activity of AsA (2 mmol/L) dissolved in SIW (66.0%) was enhanced by about 8% compared to that of the tap water (57.9%). The SOD activity of the AlEW-SIW solution (77.3%), which was higher than that of the SIW solution, and lower than that of the AlEW solution (83.6%). The SOD activity of the TMW-SIW solution (83.0%) was similar to that of the TMW solution (82.5%). Furthermore, to classify the sample solutions, discriminant analyses were performed based on near infrared (NIR) spectral data, which was consistent with the results of the WST-kit method. The SOD activity of the AlEW-SIW and TMW-SIW solutions decreased slowly with storage time, and their SOD activities were higher than that of AlEW, TMW and the tap water solutions at storage time of 14 days. In summary, AlEW-SIW and TMW-SIW showed similar antioxidant activity enhancement as AlEW and TMW, and they also maintained the stability of the antioxidant activity of AsA during storage.
Keywords: alkaline electrolyzed water; antioxidant activity; near infrared (NIR) spectroscopy; silver-ionized water; tourmaline water.
Conflict of interest statement
The authors declare no conflict of interest.
Figures







References
-
- Mahmoudi R., Aghaei S., Salehpour Z., Mousavizadeh A., Khoramrooz S.S., Taheripour Sisakht M., Christiansen G., Baneshi M., Karimi B., Bardania H. Antibacterial and antioxidant properties of phyto-synthesized silver nanoparticles using Lavandula stoechas extract. Appl. Organomet. Chem. 2020;34:e5394. doi: 10.1002/aoc.5394. - DOI
-
- Khan W., Khan N., Jamila N., Masood R., Minhaz A., Amin F., Atlas A., Nishan U. Antioxidant, antibacterial, and catalytic performance of biosynthesized silver nanoparticles of Rhus javanica, Rumex hastatus, and Callistemon viminalis. Saudi J. Biol. Sci. 2022;29:894–904. doi: 10.1016/j.sjbs.2021.10.016. - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources
Miscellaneous

