Effect-Directed, Chemical and Taxonomic Profiling of Peppermint Proprietary Varieties and Corresponding Leaf Extracts
- PMID: 36830034
- PMCID: PMC9952098
- DOI: 10.3390/antiox12020476
Effect-Directed, Chemical and Taxonomic Profiling of Peppermint Proprietary Varieties and Corresponding Leaf Extracts
Abstract
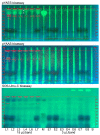
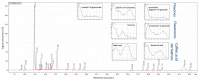
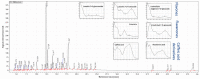
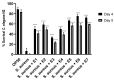
During the development of novel, standardized peppermint extracts targeting functional applications, it is critical to adequately characterize raw material plant sources to assure quality and consistency of the end-product. This study aimed to characterize existing and proprietary, newly bred varieties of peppermint and their corresponding aqueous extract products. Taxonomy was confirmed through genetic authenticity assessment. Non-target effect-directed profiling was developed using high-performance thin-layer chromatography-multi-imaging-effect-directed assays (HPTLC-UV/Vis/FLD-EDA). Results demonstrated substantial differences in compounds associated with functional attributes, notably antioxidant potential, between the peppermint samples. Further chemical analysis by high-performance liquid chromatography-photodiode array/mass spectrometry detection (HPLC-PDA/MS) and headspace solid-phase microextraction-gas chromatography-flame ionization/MS detection (headspace SPME-GC-FID/MS) confirmed compositional differences. A broad variability in the contents of flavonoids and volatiles was observed. The peppermint samples were further screened for their antioxidant potential using the Caenorhabditis elegans model, and the results indicated concordance with observed content differences of the identified functional compounds. These results documented variability among raw materials of peppermint leaves, which can yield highly variable extract products that may result in differing effects on functional targets in vivo. Hence, product standardization via effect-directed profiles is proposed as an appropriate tool.
Keywords: Caenorhabditis elegans; HPLC–PDA/MS; HPTLC–UV/Vis/FLD–EDA; antioxidant; flavonoid; functional food; headspace SPME–GC–FID/MS; high-performance thin-layer chromatography; planar bioassay; planar enzyme assay.
Conflict of interest statement
The authors declare no conflict of interest. None has received a related research grant, speaker honorarium, owns stocks, has been involved as a consultant and expert witness or is inventor of a related patent.
Figures


Similar articles
-
High-performance thin-layer chromatography linked with (bio)assays and mass spectrometry - a suited method for discovery and quantification of bioactive components? Exemplarily shown for turmeric and milk thistle extracts.J Chromatogr A. 2015 May 15;1394:137-47. doi: 10.1016/j.chroma.2015.03.029. Epub 2015 Mar 18. J Chromatogr A. 2015. PMID: 25846263
-
Forced degradation and impurity profiling: recent trends in analytical perspectives.J Pharm Biomed Anal. 2013 Dec;86:11-35. doi: 10.1016/j.jpba.2013.07.013. Epub 2013 Jul 31. J Pharm Biomed Anal. 2013. PMID: 23969330 Review.
-
Orthogonal Hyphenation of Planar and Liquid Chromatography for Mass Spectrometry of Biomarkers out of the Bioassay Matrix (NP-HPTLC-UV/vis/FLD-Bioassay-RP/IEX-HPLC-UV/vis-ESI-MS).Anal Chem. 2020 Jul 7;92(13):9057-9064. doi: 10.1021/acs.analchem.0c01251. Epub 2020 Jun 9. Anal Chem. 2020. PMID: 32452673
-
Effect-directed profiling of Ficus religiosa leaf extracts for multipotent compounds via 12 effect-directed assays.J Chromatogr A. 2021 Jan 25;1637:461836. doi: 10.1016/j.chroma.2020.461836. Epub 2020 Dec 24. J Chromatogr A. 2021. PMID: 33422795
-
Coupling of In Vitro Bioassays with Planar Chromatography in Effect-Directed Analysis.Adv Biochem Eng Biotechnol. 2017;157:187-224. doi: 10.1007/10_2016_16. Adv Biochem Eng Biotechnol. 2017. PMID: 27757476 Review.
Cited by
-
Phytochemical and Bioactivity Studies on Hedera helix L. (Ivy) Flower Pollen and Ivy Bee Pollen.Antioxidants (Basel). 2023 Jul 6;12(7):1394. doi: 10.3390/antiox12071394. Antioxidants (Basel). 2023. PMID: 37507933 Free PMC article.
-
African Under-Utilized Medicinal Leafy Vegetables Studied by Microtiter Plate Assays and High-Performance Thin-Layer Chromatography-Planar Assays.Molecules. 2024 Feb 5;29(3):733. doi: 10.3390/molecules29030733. Molecules. 2024. PMID: 38338474 Free PMC article.
-
New Antioxidant Caffeate Esters of Fatty Alcohols Identified in Robinia pseudoacacia.Molecules. 2024 Nov 30;29(23):5673. doi: 10.3390/molecules29235673. Molecules. 2024. PMID: 39683834 Free PMC article.
References
-
- Dines P.C., Falé P.L., Madeira P.J.A., Florêncio M.H., Serralheiro M.L. Acetylcholinesterase Inhibitory Activity After In Vitro Gastrointestinal Digestion of Infusions of Mentha Species. Eur. J. Med. Plants. 2012;3:381–393. doi: 10.9734/EJMP/2013/3430. - DOI
-
- Tyler V.E., Brady L.R., Robbers J.E. Pharmacognosy. 9th ed. Lea and Fabiger; Philadelphia, PA, USA: 1988.
-
- Almatroodi S.A., Alsahli M.A., Almatroudi A., Khan A.A., Rahmani A.H. Peppermint, (Mentha × piperita): Role in Management of Diseases through Modulating Various Biological Activities. Pharmacogn. J. 2021;13:822–827. doi: 10.5530/pj.2021.13.104. - DOI
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

