A Comparative Study on the Distribution Pattern of Endocrine Cells in the Gastrointestinal Tract of Two Small Alpine Mammals, Plateau Zokor (Eospalax baileyi) and Plateau Pika (Ochotona curzoniae)
- PMID: 36830427
- PMCID: PMC9951659
- DOI: 10.3390/ani13040640
A Comparative Study on the Distribution Pattern of Endocrine Cells in the Gastrointestinal Tract of Two Small Alpine Mammals, Plateau Zokor (Eospalax baileyi) and Plateau Pika (Ochotona curzoniae)
Abstract
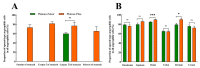
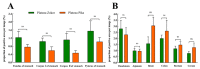
Endocrine cells can secrete a variety of gastrointestinal hormones that regulate gastrointestinal digestion and absorption, which, in turn, play an important role in animal growth, metabolism, and acclimation. The small alpine mammals, plateau zokor (Eospalax baileyi) and plateau pika (Ochotona curzoniae), live in a unique ecotope with cold, hypoxic environments and short plant-growing seasons, resulting in differential adaptive digestive strategies for foods. Studying the distribution pattern of endocrine cells in the gastrointestinal tract (GIT) of these two animals can lead to a better understanding of the survival strategies of animals in an alpine environment. In this study, we used histochemical and immunohistochemical methods to compare the distribution pattern of argyrophilic cells and the expression of 5-HT cells, Gas cells, and Glu cells in the GIT of the plateau zokor with those of the plateau pika. The results showed that these endocrine cells we studied were widely distributed in the gastrointestinal organs of both these small mammals, and their morphology and distribution location in the GIT were almost the same. However, there were significant differences in the distribution density of argyrophilic cells between different organs in the GIT. The distribution density of argyrophilic cells in the duodenum, jejunum, ileum, and rectum of plateau zokor was significantly lower than that of plateau pika (p < 0.05) and, in the cecum of plateau zokor, was significantly higher than that of plateau pika (p < 0.001). The positive expression of 5-HT cells in the corpus I, corpus II, and pylorus of the stomach, duodenum, ileum, and rectum of plateau zokor was significantly higher than that of plateau pika (p < 0.01). In addition, the positive expression of Glu cells in the cecum was significantly higher (p < 0.01) and in the duodenum and colon was significantly lower (p < 0.05) in the plateau zokor than in the plateau pika. We conclude that the distribution pattern of endocrine cells in the GIT is consistent with the respective animals' diets, with the plateau zokor feeding on high-fiber roots and plateau pika preferring to intake the aboveground parts of plants with lower fibers.
Keywords: adaptability; argyrophilic cells; gastrointestinal tract; immunoreactive cells; plateau pika; plateau zokor.
Conflict of interest statement
The authors have no conflict of interest to declare.
Figures







Similar articles
-
The Complex and Well-Developed Morphological and Histological Structures of the Gastrointestinal Tract of the Plateau Zokor Improve Its Digestive Adaptability to High-Fiber Foods.Animals (Basel). 2022 Sep 16;12(18):2447. doi: 10.3390/ani12182447. Animals (Basel). 2022. PMID: 36139307 Free PMC article.
-
[Hypoxic adaptation of the hearts of plateau zokor (Myospalax baileyi) and plateau pika (Ochotona curzoniae)].Sheng Li Xue Bao. 2008 Jun 25;60(3):348-54. Sheng Li Xue Bao. 2008. PMID: 18560725 Chinese.
-
Differences of glycolysis in skeletal muscle and lactate metabolism in liver between plateau zokor (Myospalax baileyi) and plateau pika (Ochotona curzoniae).Sheng Li Xue Bao. 2013 Jun 25;65(3):276-84. Sheng Li Xue Bao. 2013. PMID: 23788184
-
[Functional difference of malate-aspartate shuttle system in liver between plateau zokor (Myospalax baileyi) and plateau pika (Ochotona curzoniae)].Sheng Li Xue Bao. 2012 Apr 25;64(2):177-86. Sheng Li Xue Bao. 2012. PMID: 22513468 Chinese.
-
Vitamin D3 Metabolic Enzymes in Plateau Zokor (Myospalax baileyi) and Plateau Pika (Ochotona curzoniae): Expression and Response to Hypoxia.Animals (Basel). 2022 Sep 11;12(18):2371. doi: 10.3390/ani12182371. Animals (Basel). 2022. PMID: 36139230 Free PMC article.
References
-
- Meerveld G.V., Johnson A.C., Grundy D. Gastrointestinal Physiology and Function. In: Meerveld G.V., editor. Gastrointestinal Pharmacology. Volume 239. Springer; Cham, Germany: 2017. pp. 1–16. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources

