Pimozide Increases a Delayed Rectifier K+ Conductance in Chicken Embryo Vestibular Hair Cells
- PMID: 36831024
- PMCID: PMC9953418
- DOI: 10.3390/biomedicines11020488
Pimozide Increases a Delayed Rectifier K+ Conductance in Chicken Embryo Vestibular Hair Cells
Abstract
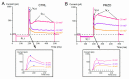
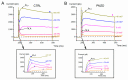
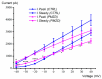
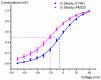
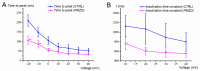
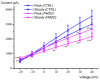
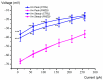
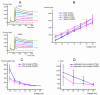
Pimozide is a conventional antipsychotic drug largely used in the therapy for schizophrenia and Tourette's syndrome. Pimozide is assumed to inhibit synaptic transmission at the CNS by acting as a dopaminergic D2 receptor antagonist. Moreover, pimozide has been shown to block voltage-gated Ca2+ and K+ channels in different cells. Despite its widespread clinical use, pimozide can cause several adverse effects, including extrapyramidal symptoms and cardiac arrhythmias. Dizziness and loss of balance are among the most common side effects of pimozide. By using the patch-clamp whole-cell technique, we investigated the effect of pimozide [3 μM] on K+ channels expressed by chicken embryo vestibular type-II hair cells. We found that pimozide slightly blocks a transient outward rectifying A-type K+ current but substantially increases a delayed outward rectifying K+ current. The net result was a significant hyperpolarization of type-II hair cells at rest and a strong reduction of their response to depolarizing stimuli. Our findings are consistent with an inhibitory effect of pimozide on the afferent synaptic transmission by type-II hair cells. Moreover, they provide an additional key to understanding the beneficial/collateral pharmacological effects of pimozide. The finding that pimozide can act as a K+ channel opener provides a new perspective for the use of this drug.
Keywords: K+ channel; balance; chicken embryo; dizziness; hair cell; ionic current; patch-clamp; pimozide; vestibular function; voltage response.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



References
-
- Rizwan M., Shahid N.U.A., Naguit N., Jakkoju R., Laeeq S., Reghefaoui T., Zahoor H., Yook J.H., Mohammed L. Efficacy of Behavioural Intervention, Antipsychotics, and Alpha Agonists in the Treatment of Tics Disorder in Tourette’s Syndrome. Cureus. 2022;14:e22449:10. doi: 10.7759/cureus.22449. - DOI - PMC - PubMed
-
- Roessner V., Eichele H., Stern J.S., Skov L., Rizzo R., Debes N.M., Nagy P., Cavanna A.E., Termine C., Ganos C., et al. European clinical guidelines for Tourette syndrome and other tic disorders—Version 2.0. Part III: Pharmacological treatment. Eur. Child Adolesc. Psychiatry. 2021;31:425–441. doi: 10.1007/s00787-021-01899-z. - DOI - PMC - PubMed
-
- Enyeart J.J., Dirksen R.T., Sharma V.K., Williford D.J., Sheu S.S. Antipsychotic pimozide is a potent Ca2+ channel blocker in heart. Mol. Pharmacol. 1990;37:752–757. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

