Cardiac-Specific Expression of Cre Recombinase Leads to Age-Related Cardiac Dysfunction Associated with Tumor-like Growth of Atrial Cardiomyocyte and Ventricular Fibrosis and Ferroptosis
- PMID: 36834504
- PMCID: PMC9962429
- DOI: 10.3390/ijms24043094
Cardiac-Specific Expression of Cre Recombinase Leads to Age-Related Cardiac Dysfunction Associated with Tumor-like Growth of Atrial Cardiomyocyte and Ventricular Fibrosis and Ferroptosis
Abstract
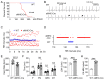
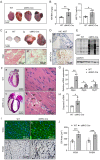
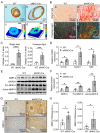
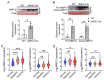
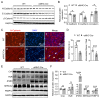
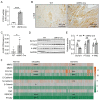
Transgenic expression of Cre recombinase driven by a specific promoter is normally used to conditionally knockout a gene in a tissue- or cell-type-specific manner. In αMHC-Cre transgenic mouse model, expression of Cre recombinase is controlled by the myocardial-specific α-myosin heavy chain (αMHC) promoter, which is commonly used to edit myocardial-specific genes. Toxic effects of Cre expression have been reported, including intro-chromosome rearrangements, micronuclei formation and other forms of DNA damage, and cardiomyopathy was observed in cardiac-specific Cre transgenic mice. However, mechanisms associated with Cardiotoxicity of Cre remain poorly understood. In our study, our data unveiled that αMHC-Cre mice developed arrhythmias and died after six months progressively, and none of them survived more than one year. Histopathological examination showed that αMHC-Cre mice had aberrant proliferation of tumor-like tissue in the atrial chamber extended from and vacuolation of ventricular myocytes. Furthermore, the αMHC-Cre mice developed severe cardiac interstitial and perivascular fibrosis, accompanied by significant increase of expression levels of MMP-2 and MMP-9 in the cardiac atrium and ventricular. Moreover, cardiac-specific expression of Cre led to disintegration of the intercalated disc, along with altered proteins expression of the disc and calcium-handling abnormality. Comprehensively, we identified that the ferroptosis signaling pathway is involved in heart failure caused by cardiac-specific expression of Cre, on which oxidative stress results in cytoplasmic vacuole accumulation of lipid peroxidation on the myocardial cell membrane. Taken together, these results revealed that cardiac-specific expression of Cre recombinase can lead to atrial mesenchymal tumor-like growth in the mice, which causes cardiac dysfunction, including cardiac fibrosis, reduction of the intercalated disc and cardiomyocytes ferroptosis at the age older than six months in mice. Our study suggests that αMHC-Cre mouse models are effective in young mice, but not in old mice. Researchers need to be particularly careful when using αMHC-Cre mouse model to interpret those phenotypic impacts of gene responses. As the Cre-associated cardiac pathology matched mostly to that of the patients, the model could also be employed for investigating age-related cardiac dysfunction.
Keywords: atrial tumors; calcium channel; cardiac-specific Cre; ferroptosis; heart failure; matrix metalloproteinases; myocardial intercalated discs.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

Similar articles
-
Cre recombinase affects calcium dynamics already in young mice.Front Pharmacol. 2025 Mar 26;16:1558573. doi: 10.3389/fphar.2025.1558573. eCollection 2025. Front Pharmacol. 2025. PMID: 40206076 Free PMC article.
-
Loss of Cardiac Ferritin H Facilitates Cardiomyopathy via Slc7a11-Mediated Ferroptosis.Circ Res. 2020 Jul 31;127(4):486-501. doi: 10.1161/CIRCRESAHA.120.316509. Epub 2020 Apr 30. Circ Res. 2020. PMID: 32349646
-
Conditional ablation of nonmuscle myosin II-B delineates heart defects in adult mice.Circ Res. 2009 Nov 20;105(11):1102-9. doi: 10.1161/CIRCRESAHA.109.200303. Epub 2009 Oct 8. Circ Res. 2009. PMID: 19815823 Free PMC article.
-
The Role of the L-Type Ca2+ Channel in Altered Metabolic Activity in a Murine Model of Hypertrophic Cardiomyopathy.JACC Basic Transl Sci. 2016 Feb 13;1(1-2):61-72. doi: 10.1016/j.jacbts.2015.12.001. eCollection 2016 Jan-Feb. JACC Basic Transl Sci. 2016. PMID: 30167506 Free PMC article. Review.
-
Ferroptosis and its role in cardiomyopathy.Biomed Pharmacother. 2022 Sep;153:113279. doi: 10.1016/j.biopha.2022.113279. Epub 2022 Jun 20. Biomed Pharmacother. 2022. PMID: 35738177 Review.
Cited by
-
MicroRNA205: A Key Regulator of Cardiomyocyte Transition from Proliferative to Hypertrophic Growth in the Neonatal Heart.Int J Mol Sci. 2024 Feb 12;25(4):2206. doi: 10.3390/ijms25042206. Int J Mol Sci. 2024. PMID: 38396885 Free PMC article.
-
Identification of ferroptosis biomarkers and immune infiltration landscapes in atrial fibrillation: A bioinformatics analysis.Medicine (Baltimore). 2024 Sep 27;103(39):e39777. doi: 10.1097/MD.0000000000039777. Medicine (Baltimore). 2024. PMID: 39331874 Free PMC article.
-
Cre recombinase affects calcium dynamics already in young mice.Front Pharmacol. 2025 Mar 26;16:1558573. doi: 10.3389/fphar.2025.1558573. eCollection 2025. Front Pharmacol. 2025. PMID: 40206076 Free PMC article.
-
Mechanisms underlying dilated cardiomyopathy associated with FKBP12 deficiency.J Gen Physiol. 2025 Jan 6;157(1):e202413583. doi: 10.1085/jgp.202413583. Epub 2024 Dec 11. J Gen Physiol. 2025. PMID: 39661086 Free PMC article.
-
The role of CYP-sEH derived lipid mediators in regulating mitochondrial biology and cellular senescence: implications for the aging heart.Front Pharmacol. 2024 Dec 5;15:1486717. doi: 10.3389/fphar.2024.1486717. eCollection 2024. Front Pharmacol. 2024. PMID: 39703395 Free PMC article. Review.
References
-
- Sauer B., Henderson N. Targeted insertion of exogenous DNA into the eukaryotic genome by the Cre recombinase. New Biol. 1990;2:441–449. - PubMed
MeSH terms
Substances
Grants and funding
- 31771377 / 31571273 / 31371256/the National Natural Science Foundation of China
- MS2014SXSF038/the Foreign Distinguished Scientist Program from the National Department of Education
- GK201301001/ 201701005/GERP-17-45/the National Department of Education Central Universities Research Fund
- GK202207004/the National Department of Education Central Universities Research Fund
- 2019TS079/Advanced Cell Biology for Graduated Study
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

