New Insights into the Regulation of mTOR Signaling via Ca2+-Binding Proteins
- PMID: 36835331
- PMCID: PMC9959742
- DOI: 10.3390/ijms24043923
New Insights into the Regulation of mTOR Signaling via Ca2+-Binding Proteins
Abstract
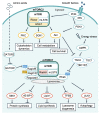
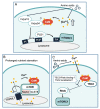
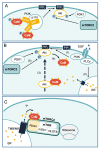
Environmental factors are important regulators of cell growth and proliferation. Mechanistic target of rapamycin (mTOR) is a central kinase that maintains cellular homeostasis in response to a variety of extracellular and intracellular inputs. Dysregulation of mTOR signaling is associated with many diseases, including diabetes and cancer. Calcium ion (Ca2+) is important as a second messenger in various biological processes, and its intracellular concentration is tightly regulated. Although the involvement of Ca2+ mobilization in mTOR signaling has been reported, the detailed molecular mechanisms by which mTOR signaling is regulated are not fully understood. The link between Ca2+ homeostasis and mTOR activation in pathological hypertrophy has heightened the importance in understanding Ca2+-regulated mTOR signaling as a key mechanism of mTOR regulation. In this review, we introduce recent findings on the molecular mechanisms of regulation of mTOR signaling by Ca2+-binding proteins, particularly calmodulin (CaM).
Keywords: Ca2+ mobilization; TSC2; calmodulin; mTOR.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
Similar articles
-
hsBAFF promotes proliferation and survival in cultured B lymphocytes via calcium signaling activation of mTOR pathway.Cytokine. 2013 May;62(2):310-21. doi: 10.1016/j.cyto.2013.03.011. Epub 2013 Apr 1. Cytokine. 2013. PMID: 23557796
-
Amino acids activate mTOR complex 1 via Ca2+/CaM signaling to hVps34.Cell Metab. 2008 May;7(5):456-65. doi: 10.1016/j.cmet.2008.03.002. Cell Metab. 2008. PMID: 18460336 Free PMC article.
-
Ca2+/Calmodulin induces translocation of membrane-associated TSC2 to the nucleus where it suppresses CYP24A1 expression.Biosci Biotechnol Biochem. 2022 Dec 21;87(1):45-53. doi: 10.1093/bbb/zbac174. Biosci Biotechnol Biochem. 2022. PMID: 36331254
-
TOR Signaling Pathway in Cardiac Aging and Heart Failure.Biomolecules. 2021 Jan 27;11(2):168. doi: 10.3390/biom11020168. Biomolecules. 2021. PMID: 33513917 Free PMC article. Review.
-
mTOR and regulation of energy homeostasis in humans.J Mol Med (Berl). 2013 Oct;91(10):1167-75. doi: 10.1007/s00109-013-1057-6. Epub 2013 Jun 12. J Mol Med (Berl). 2013. PMID: 23756767 Review.
Cited by
-
From Mechanoelectric Conversion to Tissue Regeneration: Translational Progress in Piezoelectric Materials.Adv Mater. 2025 Aug;37(33):e2417564. doi: 10.1002/adma.202417564. Epub 2025 May 28. Adv Mater. 2025. PMID: 40434211 Free PMC article. Review.
-
mTORC1 and SGLT2 Inhibitors-A Therapeutic Perspective for Diabetic Cardiomyopathy.Int J Mol Sci. 2023 Oct 11;24(20):15078. doi: 10.3390/ijms242015078. Int J Mol Sci. 2023. PMID: 37894760 Free PMC article. Review.
-
The mTORC2 signaling network: targets and cross-talks.Biochem J. 2024 Jan 25;481(2):45-91. doi: 10.1042/BCJ20220325. Biochem J. 2024. PMID: 38270460 Free PMC article. Review.
-
Transplantation of dorsal root ganglia overexpressing the NaChBac sodium channel improves locomotion after complete SCI.Mol Ther. 2024 Jun 5;32(6):1739-1759. doi: 10.1016/j.ymthe.2024.03.038. Epub 2024 Mar 30. Mol Ther. 2024. PMID: 38556794 Free PMC article.
-
Calmodulin enhances mTORC1 signaling by preventing TSC2-Rheb binding.J Biol Chem. 2025 Feb;301(2):108122. doi: 10.1016/j.jbc.2024.108122. Epub 2024 Dec 22. J Biol Chem. 2025. PMID: 39716490 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

