Benefit of Adjuvant Mesenchymal Stem Cell Transplantation to Critical-Sized Peripheral Nerve Defect Repair: A Systematic Review and Meta-Analysis of Preclinical Studies
- PMID: 36835844
- PMCID: PMC9966712
- DOI: 10.3390/jcm12041306
Benefit of Adjuvant Mesenchymal Stem Cell Transplantation to Critical-Sized Peripheral Nerve Defect Repair: A Systematic Review and Meta-Analysis of Preclinical Studies
Abstract
Critically sized nerve defects cause devastating life-long disabilities and require interposition for reconstruction. Additional local application of mesenchymal stem cells (MSCs) is considered promising to enhance peripheral nerve regeneration. To better understand the role of MSCs in peripheral nerve reconstruction, we performed a systematic review and meta-analysis of the effects of MSCs on critically sized segment nerve defects in preclinical studies. 5146 articles were screened following PRISMA guidelines using PubMed and Web of Science. A total of 27 preclinical studies (n = 722 rats) were included in the meta-analysis. The mean difference or the standardized mean difference with 95% confidence intervals for motor function, conduction velocity, and histomorphological parameters of nerve regeneration, as well as the degree of muscle atrophy, was compared in rats with critically sized defects and autologous nerve reconstruction treated with or without MSCs. The co-transplantation of MSCs increased the sciatic functional index (3.93, 95% CI 2.62 to 5.24, p < 0.00001) and nerve conduction velocity recovery (1.49, 95% CI 1.13 to 1.84, p = 0.009), decreased the atrophy of targeted muscles (gastrocnemius: 0.63, 95% CI 0.29 to 0.97 p = 0.004; triceps surae: 0.08, 95% CI 0.06 to 0.10 p = 0.71), and promoted the regeneration of injured axons (axon number: 1.10, 95% CI 0.78 to 1.42, p < 0.00001; myelin sheath thickness: 0.15, 95% CI 0.12 to 0.17, p = 0.28). Reconstruction of critically sized peripheral nerve defects is often hindered by impaired postoperative regeneration, especially in defects that require an autologous nerve graft. This meta-analysis indicates that additional application of MSC can enhance postoperative peripheral nerve regeneration in rats. Based on the promising results in vivo experiments, further studies are needed to demonstrate potential clinical benefits.
Keywords: MSC; nerve injury; nerve reconstruction; peripheral nerve; stem cell; trauma.
Conflict of interest statement
None of the authors has a potential conflict of interest with respect to the research, authorship, mentioned products or devices, and publication of this article.
Figures
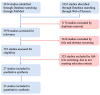
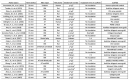
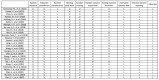





References
-
- Aman M., Zimmermann K.S., Thielen M., Thomas B., Daeschler S., Boecker A.H., Stolle A., Bigdeli A.K., Kneser U., Harhaus L. An Epidemiological and Etiological Analysis of 5026 Peripheral Nerve Lesions from a European Level I Trauma Center. J. Pers. Med. 2022;12:1673. doi: 10.3390/jpm12101673. - DOI - PMC - PubMed
-
- Siemionow M., Strojny M.M., Kozlowska K., Brodowska S., Grau-Kazmierczak W., Cwykiel J. Application of Human Epineural Conduit Supported with Human Mesenchymal Stem Cells as a Novel Therapy for Enhancement of Nerve Gap Regeneration. Stem Cell Rev. Rep. 2021;18:642–659. doi: 10.1007/s12015-021-10301-z. - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources

