ESKD Risk Prediction Model in a Multicenter Chronic Kidney Disease Cohort in China: A Derivation, Validation, and Comparison Study
- PMID: 36836039
- PMCID: PMC9965616
- DOI: 10.3390/jcm12041504
ESKD Risk Prediction Model in a Multicenter Chronic Kidney Disease Cohort in China: A Derivation, Validation, and Comparison Study
Abstract
Background and objectives: In light of the growing burden of chronic kidney disease (CKD), it is of particular importance to create disease prediction models that can assist healthcare providers in identifying cases of CKD individual risk and integrate risk-based care for disease progress management. The objective of this study was to develop and validate a new pragmatic end-stage kidney disease (ESKD) risk prediction utilizing the Cox proportional hazards model (Cox) and machine learning (ML).
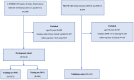
Design, setting, participants, and measurements: The Chinese Cohort Study of Chronic Kidney Disease (C-STRIDE), a multicenter CKD cohort in China, was employed as the model's training and testing datasets, with a split ratio of 7:3. A cohort from Peking University First Hospital (PKUFH cohort) served as the external validation dataset. The participants' laboratory tests in those cohorts were conducted at PKUFH. We included individuals with CKD stages 1~4 at baseline. The incidence of kidney replacement therapy (KRT) was defined as the outcome. We constructed the Peking University-CKD (PKU-CKD) risk prediction model employing the Cox and ML methods, which include extreme gradient boosting (XGBoost) and survival support vector machine (SSVM). These models discriminate metrics by applying Harrell's concordance index (Harrell's C-index) and Uno's concordance (Uno's C). The calibration performance was measured by the Brier score and plots.
Results: Of the 3216 C-STRIDE and 342 PKUFH participants, 411 (12.8%) and 25 (7.3%) experienced KRT with mean follow-up periods of 4.45 and 3.37 years, respectively. The features included in the PKU-CKD model were age, gender, estimated glomerular filtration rate (eGFR), urinary albumin-creatinine ratio (UACR), albumin, hemoglobin, medical history of type 2 diabetes mellitus (T2DM), and hypertension. In the test dataset, the values of the Cox model for Harrell's C-index, Uno's C-index, and Brier score were 0.834, 0.833, and 0.065, respectively. The XGBoost algorithm values for these metrics were 0.826, 0.825, and 0.066, respectively. The SSVM model yielded values of 0.748, 0.747, and 0.070, respectively, for the above parameters. The comparative analysis revealed no significant difference between XGBoost and Cox, in terms of Harrell's C, Uno's C, and the Brier score (p = 0.186, 0.213, and 0.41, respectively) in the test dataset. The SSVM model was significantly inferior to the previous two models (p < 0.001), in terms of discrimination and calibration. The validation dataset showed that XGBoost was superior to Cox, regarding Harrell's C, Uno's C, and the Brier score (p = 0.003, 0.027, and 0.032, respectively), while Cox and SSVM were almost identical concerning these three parameters (p = 0.102, 0.092, and 0.048, respectively).
Conclusions: We developed and validated a new ESKD risk prediction model for patients with CKD, employing commonly measured indicators in clinical practice, and its overall performance was satisfactory. The conventional Cox regression and certain ML models exhibited equal accuracy in predicting the course of CKD.
Keywords: chronic kidney disease; machine learning; prediction model; progression.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
Similar articles
-
Development and Validation of a Machine Learning-Based Prognostic Model for IgA Nephropathy with Chronic Kidney Disease Stage 3 or 4.Kidney Dis (Basel). 2024 Aug 22;10(6):436-449. doi: 10.1159/000540682. eCollection 2024 Dec. Kidney Dis (Basel). 2024. PMID: 39664336 Free PMC article.
-
Interpretable machine learning for predicting chronic kidney disease progression risk.Digit Health. 2024 Jan 15;10:20552076231224225. doi: 10.1177/20552076231224225. eCollection 2024 Jan-Dec. Digit Health. 2024. PMID: 38235416 Free PMC article.
-
Development and validation of a risk prediction model for chronic kidney disease among individuals with type 2 diabetes.Sci Rep. 2022 Mar 21;12(1):4794. doi: 10.1038/s41598-022-08284-z. Sci Rep. 2022. PMID: 35314714 Free PMC article. Clinical Trial.
-
Performance of prediction models for nephropathy in people with type 2 diabetes: systematic review and external validation study.BMJ. 2021 Sep 28;374:n2134. doi: 10.1136/bmj.n2134. BMJ. 2021. PMID: 34583929 Free PMC article.
-
Modeling Chronic Kidney Disease in Type 2 Diabetes Mellitus: A Systematic Literature Review of Models, Data Sources, and Derivation Cohorts.Diabetes Ther. 2022 Apr;13(4):651-677. doi: 10.1007/s13300-022-01208-0. Epub 2022 Mar 15. Diabetes Ther. 2022. PMID: 35290625 Free PMC article. Review.
Cited by
-
A Klotho-Based Machine Learning Model for Prediction of both Kidney and Cardiovascular Outcomes in Chronic Kidney Disease.Kidney Dis (Basel). 2024 Mar 25;10(3):200-212. doi: 10.1159/000538510. eCollection 2024 Jun. Kidney Dis (Basel). 2024. PMID: 38835404 Free PMC article.
-
Revolutionizing Chronic Kidney Disease Management with Machine Learning and Artificial Intelligence.J Clin Med. 2023 Apr 21;12(8):3018. doi: 10.3390/jcm12083018. J Clin Med. 2023. PMID: 37109354 Free PMC article.
-
A SuperLearner approach for predicting diabetic kidney disease upon the initial diagnosis of T2DM in hospital.BMC Med Inform Decis Mak. 2025 Mar 26;25(1):148. doi: 10.1186/s12911-025-02977-x. BMC Med Inform Decis Mak. 2025. PMID: 40140809 Free PMC article.
-
Development, validation and economic evaluation of a machine learning algorithm for predicting the probability of kidney damage in patients with hyperuricaemia: protocol for a retrospective study.BMJ Open. 2024 Nov 28;14(11):e086032. doi: 10.1136/bmjopen-2024-086032. BMJ Open. 2024. PMID: 39613447 Free PMC article.
-
Dynamic survival prediction of end-stage kidney disease using random survival forests for competing risk analysis.Front Med (Lausanne). 2024 Dec 11;11:1428073. doi: 10.3389/fmed.2024.1428073. eCollection 2024. Front Med (Lausanne). 2024. PMID: 39722823 Free PMC article.
References
-
- Bikbov B., Purcell C.A., Levey A.S., Smith M., Abdoli A., Abebe M., Owolabi M.O. Global, regional, and national burden of chronic kidney disease, 1990–2017: A systematic analysis for the Global Burden of Disease Study 2017. Lancet. 2020;395:709–733. doi: 10.1016/S0140-6736(20)30045-3. - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

