Association between Gut Microbiota and SARS-CoV-2 Infection and Vaccine Immunogenicity
- PMID: 36838417
- PMCID: PMC9961186
- DOI: 10.3390/microorganisms11020452
Association between Gut Microbiota and SARS-CoV-2 Infection and Vaccine Immunogenicity
Abstract
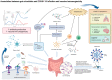
Gut microbiota is increasingly recognized to play a pivotal role in various human physiological functions and diseases. Amidst the COVID-19 pandemic, research has suggested that dysbiosis of the gut microbiota is also involved in the development and severity of COVID-19 symptoms by regulating SARS-CoV-2 entry and modulating inflammation. Previous studies have also suggested that gut microbiota and their metabolites could have immunomodulatory effects on vaccine immunogenicity, including influenza vaccines and oral rotavirus vaccines. In light of these observations, it is possible that gut microbiota plays a role in influencing the immune responses to COVID-19 vaccinations via similar mechanisms including effects of lipopolysaccharides, flagellin, peptidoglycan, and short-chain fatty acids. In this review, we give an overview of the current understanding on the role of the gut microbiota in COVID-19 manifestations and vaccine immunogenicity. We then discuss the limitations of currently published studies on the associations between gut microbiota and COVID-19 vaccine outcomes. Future research directions shall be focused on the development of microbiota-based interventions on improving immune response to SARS-CoV-2 infection and vaccinations.
Keywords: COVID-19; COVID-19 vaccine; gut dysbiosis; gut microbiota; vaccine immunogenicity.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
Similar articles
-
Dietary intervention with functional foods modulating gut microbiota for improving the efficacy of COVID-19 vaccines.Heliyon. 2023 May;9(5):e15668. doi: 10.1016/j.heliyon.2023.e15668. Epub 2023 Apr 21. Heliyon. 2023. PMID: 37124341 Free PMC article. Review.
-
The intestinal microbiota and improving the efficacy of COVID-19 vaccinations.J Funct Foods. 2021 Dec;87:104850. doi: 10.1016/j.jff.2021.104850. Epub 2021 Nov 10. J Funct Foods. 2021. PMID: 34777578 Free PMC article. Review.
-
Current Drug Targets for Gut Microbiota Biocorrection during the SARS-CoV-2 Pandemic: A Systematic Review.Curr Drug Targets. 2022;23(11):1099-1125. doi: 10.2174/1389450123666220418094853. Curr Drug Targets. 2022. PMID: 35440305
-
Alterations of the gut microbiota in coronavirus disease 2019 and its therapeutic potential.World J Gastroenterol. 2022 Dec 21;28(47):6689-6701. doi: 10.3748/wjg.v28.i47.6689. World J Gastroenterol. 2022. PMID: 36620345 Free PMC article. Review.
-
Roles of the gut microbiota in severe SARS-CoV-2 infection.Cytokine Growth Factor Rev. 2022 Feb;63:98-107. doi: 10.1016/j.cytogfr.2022.01.007. Epub 2022 Jan 31. Cytokine Growth Factor Rev. 2022. PMID: 35131164 Free PMC article. Review.
Cited by
-
The Intersection of COVID-19 and Metabolic-Associated Fatty Liver Disease: An Overview of the Current Evidence.Viruses. 2023 Apr 27;15(5):1072. doi: 10.3390/v15051072. Viruses. 2023. PMID: 37243158 Free PMC article. Review.
-
Effect of metabolic dysfunction-associated steatotic liver disease on BNT162b2 immunogenicity against the severe acute respiratory syndrome coronavirus 2 omicron variant.J Gastroenterol Hepatol. 2024 Nov;39(11):2386-2393. doi: 10.1111/jgh.16716. Epub 2024 Aug 17. J Gastroenterol Hepatol. 2024. PMID: 39152762 Free PMC article.
-
Baseline Gut Microbiota Was Associated with Long-Term Immune Response at One Year Following Three Doses of BNT162b2.Vaccines (Basel). 2024 Aug 14;12(8):916. doi: 10.3390/vaccines12080916. Vaccines (Basel). 2024. PMID: 39204040 Free PMC article.
-
Alterations in microbiota of patients with COVID-19: implications for therapeutic interventions.MedComm (2020). 2024 Mar 15;5(4):e513. doi: 10.1002/mco2.513. eCollection 2024 Apr. MedComm (2020). 2024. PMID: 38495122 Free PMC article. Review.
-
Unraveling the Impact of Gut and Oral Microbiome on Gut Health in Inflammatory Bowel Diseases.Nutrients. 2023 Jul 29;15(15):3377. doi: 10.3390/nu15153377. Nutrients. 2023. PMID: 37571313 Free PMC article. Review.
References
-
- Cheung K.S., Hung I.F.N., Chan P.P.Y., Lung K.C., Tso E., Liu R., Ng Y.Y., Chu M.Y., Chung T.W.H., Tam A.R., et al. Gastrointestinal Manifestations of SARS-CoV-2 Infection and Virus Load in Fecal Samples from a Hong Kong Cohort: Systematic Review and Meta-analysis. Gastroenterology. 2020;159:81–95. doi: 10.1053/j.gastro.2020.03.065. - DOI - PMC - PubMed
-
- Mao R., Qiu Y., He J.S., Tan J.-Y., Li X.-H., Liang J., Shen J., Zhu L.-R., Chen Y., Iacucci M., et al. Manifestations and prognosis of gastrointestinal and liver involvement in patients with COVID-19: A systematic review and meta-analysis. Lancet Gastroenterol. Hepatol. 2020;5:667–678. doi: 10.1016/S2468-1253(20)30126-6. - DOI - PMC - PubMed
-
- Pasolli E., Asnicar F., Manara S., Zolfo M., Karcher N., Armanini F., Beghini F., Manghi P., Tett A., Ghensi P., et al. Extensive Unexplored Human Microbiome Diversity Revealed by Over 150,000 Genomes from Metagenomes Spanning Age, Geography, and Lifestyle. Cell. 2019;176:649–662.e20. doi: 10.1016/j.cell.2019.01.001. - DOI - PMC - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Miscellaneous

