Regulation of Reactive Oxygen Species during Salt Stress in Plants and Their Crosstalk with Other Signaling Molecules-Current Perspectives and Future Directions
- PMID: 36840211
- PMCID: PMC9964777
- DOI: 10.3390/plants12040864
Regulation of Reactive Oxygen Species during Salt Stress in Plants and Their Crosstalk with Other Signaling Molecules-Current Perspectives and Future Directions
Abstract
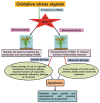
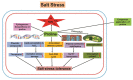
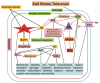
Salt stress is a severe type of environmental stress. It adversely affects agricultural production worldwide. The overproduction of reactive oxygen species (ROS) is the most frequent phenomenon during salt stress. ROS are extremely reactive and, in high amounts, noxious, leading to destructive processes and causing cellular damage. However, at lower concentrations, ROS function as secondary messengers, playing a critical role as signaling molecules, ensuring regulation of growth and adjustment to multifactorial stresses. Plants contain several enzymatic and non-enzymatic antioxidants that can detoxify ROS. The production of ROS and their scavenging are important aspects of the plant's normal response to adverse conditions. Recently, this field has attracted immense attention from plant scientists; however, ROS-induced signaling pathways during salt stress remain largely unknown. In this review, we will discuss the critical role of different antioxidants in salt stress tolerance. We also summarize the recent advances on the detrimental effects of ROS, on the antioxidant machinery scavenging ROS under salt stress, and on the crosstalk between ROS and other various signaling molecules, including nitric oxide, hydrogen sulfide, calcium, and phytohormones. Moreover, the utilization of "-omic" approaches to improve the ROS-regulating antioxidant system during the adaptation process to salt stress is also described.
Keywords: ascorbate peroxidase; catalase; hydrogen sulfide; nitric oxide; omics; phytohormones; reactive oxygen species; salt stress; superoxide dismutase.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



References
-
- Kesawat M.S., Kherawat B.S., Singh A., Dey P., Kabi M., Debnath D., Saha D., Khandual A., Rout S., Ali A. Genome-wide identification and characterization of the brassinazole-resistant (BZR) gene family and its expression in the various developmental stage and stress conditions in wheat (Triticum aestivum L.) Int. J. Mol. Sci. 2021;22:8743. doi: 10.3390/ijms22168743. - DOI - PMC - PubMed
-
- Kesawat M.S., Kherawat B.S., Singh A., Dey P., Routray S., Mohapatra C., Saha D., Ram C., Siddique K.H., Kumar A. Genome-Wide Analysis and Characterization of the Proline-Rich Extensin-like Receptor Kinases (PERKs) Gene Family Reveals Their Role in Different Developmental Stages and Stress Conditions in Wheat (Triticum aestivum L.) Plants. 2022;11:496. doi: 10.3390/plants11040496. - DOI - PMC - PubMed
Publication types
LinkOut - more resources
Full Text Sources

