The accelerated waning of immunity and reduced effect of booster in patients treated with bDMARD and tsDMARD after SARS-CoV-2 mRNA vaccination
- PMID: 36844197
- PMCID: PMC9947701
- DOI: 10.3389/fmed.2023.1049157
The accelerated waning of immunity and reduced effect of booster in patients treated with bDMARD and tsDMARD after SARS-CoV-2 mRNA vaccination
Abstract
Objectives: This study aimed to assess the duration of humoral responses after two doses of SARS-CoV-2 mRNA vaccines in patients with inflammatory joint diseases and IBD and booster vaccination compared with healthy controls. It also aimed to analyze factors influencing the quantity and quality of the immune response.
Methods: We enrolled 41 patients with rheumatoid arthritis (RA), 35 with seronegative spondyloarthritis (SpA), and 41 suffering from inflammatory bowel disease (IBD), excluding those receiving B-cell-depleting therapies. We assessed total anti-SARS-CoV-2 spike antibodies (Abs) and neutralizing Ab titers 6 months after two and then after three doses of mRNA vaccines compared with healthy controls. We analyzed the influence of therapies on the humoral response.
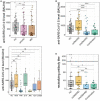
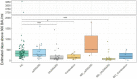
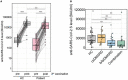
Results: Patients receiving biological or targeted synthetic disease-modifying antirheumatic drugs (b/tsDMARDs) showed reduced anti-SARS-CoV-2 S Abs and neutralizing Ab titers compared with HC or patients receiving conventional synthetic (cs)DMARDs 6 months after the first two vaccination doses. Anti-SARS-CoV-2 S titers of patients with b/tsDMARDs declined more rapidly, leading to a significant reduction in the duration of vaccination-induced immunity after two doses of SARS-CoV-2 mRNA vaccines. While 23% of HC and 19% of patients receiving csDMARDs were without detectable neutralizing Abs 6 months after the first two vaccination doses, this number was 62% in patients receiving b/tsDMARDs and 52% in patients receiving a combination of csDMARDs and b/tsDMARDs. Booster vaccination led to increased anti-SARS-CoV-2 S Abs in all HC and patients. However, anti-SARS-CoV-2 S Abs after booster vaccination was diminished in patients receiving b/tsDMARDs, either alone or in combination with csDMARDs compared to HC.
Conclusion: Patients receiving b/tsDMARDs have significantly reduced Abs and neutralizing Ab titers 6 months after mRNA vaccination against SARS-CoV-2. This was due to a faster decline in Ab levels, indicating a significantly reduced duration of vaccination-induced immunity compared with HC or patients receiving csDMARDs. In addition, they display a reduced response to a booster vaccination, warranting earlier booster vaccination strategies in patients under b/tsDMARD therapy, according to their specific Ab levels.
Keywords: IBD; SARS-CoV-2; arthritis; bDMARD; humoral immune response; immunomodulatory therapy.
Copyright © 2023 Tobudic, Simader, Deimel, Straub, Kartnig, Heinz, Mandl, Haslacher, Perkmann, Schneider, Nothnagl, Radner, Winkler, Burgmann, Stiasny, Novacek, Reinisch, Aletaha, Winkler and Blüml.
Conflict of interest statement
PM reports speaker fees from AbbVie, Janssen and Novartis and research grants from AbbVie, BMS, Novartis, Janssen, MSD, and UCB outside of the submitted work. KS reports a research grant from Pfizer, outside the submitted work. HH received grants from Glock Health, BlueSky Immunotherapies, and Neutrolis. HB received consulting fees from MSD, Pfizer, Takeda, Gilead, speaker fees from Shionogi, Pfizer, MSD, advisory boards for Valneva, MSD, Gilead. DA reports grants from Abbvie, Amgen, Lilly, Novartis, Roche, SoBi, Sanofi, other from Abbvie, Amgen, Lilly, Merck, Novartis, Pfizer, Roche, Sandoz, outside the submitted work. SB reports personal fees from Abbvie, personal fees from Novartis, outside the submitted work. ES reports supports for attendance of meetings from Pfizer, Bristol-Myers Squibb, Boehringer Ingelheim. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures

References
-
- Folegatti PM, Ewer KJ, Aley PK, Angus B, Becker S, Belij-Rammerstorfer S, et al. Safety and immunogenicity of the ChAdOx1 nCoV-19 vaccine against SARS-CoV-2: a preliminary report of a phase 1/2, single-blind, randomised controlled trial. Lancet. (2020) 396:467–78. 10.1016/S0140-6736(20)31604-4 - DOI - PMC - PubMed
-
- Ramasamy MN, Minassian AM, Ewer KJ, Flaxman AL, Folegatti PM, Owens DR, et al. Safety and immunogenicity of ChAdOx1 nCoV-19 vaccine administered in a prime-boost regimen in young and old adults (COV002): a single-blind, randomised, controlled, phase 2/3 trial. Lancet. (2021) 396:1979–93. 10.1016/S0140-6736(20)32466-1 - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources
Miscellaneous

