Advancing personalized medicine for tuberculosis through the application of immune profiling
- PMID: 36844400
- PMCID: PMC9950414
- DOI: 10.3389/fcimb.2023.1108155
Advancing personalized medicine for tuberculosis through the application of immune profiling
Abstract
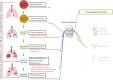
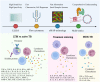
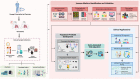
While early and precise diagnosis is the key to eliminating tuberculosis (TB), conventional methods using culture conversion or sputum smear microscopy have failed to meet demand. This is especially true in high-epidemic developing countries and during pandemic-associated social restrictions. Suboptimal biomarkers have restricted the improvement of TB management and eradication strategies. Therefore, the research and development of new affordable and accessible methods are required. Following the emergence of many high-throughput quantification TB studies, immunomics has the advantages of directly targeting responsive immune molecules and significantly simplifying workloads. In particular, immune profiling has been demonstrated to be a versatile tool that potentially unlocks many options for application in TB management. Herein, we review the current approaches for TB control with regard to the potentials and limitations of immunomics. Multiple directions are also proposed to hopefully unleash immunomics' potential in TB research, not least in revealing representative immune biomarkers to correctly diagnose TB. The immune profiles of patients can be valuable covariates for model-informed precision dosing-based treatment monitoring, prediction of outcome, and the optimal dose prediction of anti-TB drugs.
Keywords: biomarkers; immunomics; model-informed precision dosing; multi-omics; personalized medicine; tuberculosis.
Copyright © 2023 Thu, Dat, Jayanti, Trinh, Hung, Cho, Long and Shin.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures


References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

