Functional characterization of TSPAN7 as a novel indicator for immunotherapy in glioma
- PMID: 36845098
- PMCID: PMC9947846
- DOI: 10.3389/fimmu.2023.1105489
Functional characterization of TSPAN7 as a novel indicator for immunotherapy in glioma
Abstract
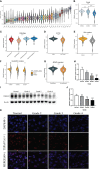
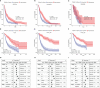
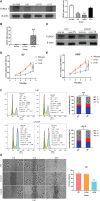
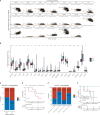
Glioma is the most common primary malignant tumor of the central nervous system in clinical practice. Most adult diffuse gliomas have poor efficacy after standard treatment, especially glioblastoma. With the in-depth understanding of brain immune microenvironment, immunotherapy as a new treatment has attracted much attention. In this study, through analyzing a large number of glioma cohorts, we reported that TSPAN7, a member of the tetraspanin family, decreased in high-grade gliomas and low expression was associated with poor prognosis in glioma patients. Meanwhile, the expression pattern of TSPAN7 was verified in glioma clinical samples and glioma cell lines by qPCR, Western Blotting and immunofluorescence. In addition, functional enrichment analysis showed that cell proliferation, EMT, angiogenesis, DNA repair and MAPK signaling pathways were activated in the TSPAN7 lower expression subgroup. Lentiviral plasmids were used to overexpress TSPAN7 in U87 and LN229 glioma cell lines to explore the anti-tumor role of TSPAN7 in glioma. Moreover, by analyzing the relationship between TSPAN7 expression and immune cell infiltration in multiple datasets, we found that TSPAN7 was significantly negatively correlated with the immune infiltration of tumor-related macrophages, especially M2-type macrophages. Further analysis of immune checkpoints showed that, the expression level of TSPAN7 was negatively correlated with the expression of PD-1, PD-L1 and CTLA-4. Using an independent anti-PD-1 immunotherapy cohorts of GBM, we demonstrated that TSPAN7 expression may had a synergistic effect with PD-L1 on the response to immunotherapy. Based on the above findings, we speculate that TSPAN7 can serve as a biomarker for prognosis and a potential immunotherapy target in glioma patients.
Keywords: glioma; immune infiltration; immunotherapy; malignant progression; single-cell RNA sequencing.
Copyright © 2023 Chen, Liu, Li, Lin, Xia, Wanggou and Li.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest, and no conflicts of interest exist among the authors.
Figures



Similar articles
-
MET overexpression contributes to STAT4-PD-L1 signaling activation associated with tumor-associated, macrophages-mediated immunosuppression in primary glioblastomas.J Immunother Cancer. 2021 Oct;9(10):e002451. doi: 10.1136/jitc-2021-002451. J Immunother Cancer. 2021. PMID: 34667077 Free PMC article.
-
Enhanced B7-H4 expression in gliomas with low PD-L1 expression identifies super-cold tumors.J Immunother Cancer. 2020 May;8(1):e000154. doi: 10.1136/jitc-2019-000154. J Immunother Cancer. 2020. PMID: 32457124 Free PMC article.
-
Orphan nuclear receptor TLX promotes immunosuppression via its transcriptional activation of PD-L1 in glioma.J Immunother Cancer. 2021 Apr;9(4):e001937. doi: 10.1136/jitc-2020-001937. J Immunother Cancer. 2021. PMID: 33858847 Free PMC article.
-
A Systematic Review of the Tumor-Infiltrating CD8+ T-Cells/PD-L1 Axis in High-Grade Glial Tumors: Toward Personalized Immuno-Oncology.Front Immunol. 2021 Sep 17;12:734956. doi: 10.3389/fimmu.2021.734956. eCollection 2021. Front Immunol. 2021. PMID: 34603316 Free PMC article.
-
Current advances in PD-1/PD-L1 axis-related tumour-infiltrating immune cells and therapeutic regimens in glioblastoma.Crit Rev Oncol Hematol. 2020 Jul;151:102965. doi: 10.1016/j.critrevonc.2020.102965. Epub 2020 Apr 24. Crit Rev Oncol Hematol. 2020. PMID: 32442903 Review.
Cited by
-
Tetraspanin7 in adipose tissue remodeling and its impact on metabolic health.Mol Metab. 2025 Jul;97:102168. doi: 10.1016/j.molmet.2025.102168. Epub 2025 May 12. Mol Metab. 2025. PMID: 40368161 Free PMC article.
-
Unveiling the unique role of TSPAN7 across tumors: a pan-cancer study incorporating retrospective clinical research and bioinformatic analysis.Biol Direct. 2024 Aug 22;19(1):72. doi: 10.1186/s13062-024-00516-8. Biol Direct. 2024. PMID: 39175035 Free PMC article.
-
Glioblastoma-associated macrophages in glioblastoma: from their function and mechanism to therapeutic advances.Cancer Gene Ther. 2025 Jun;32(6):595-607. doi: 10.1038/s41417-025-00905-9. Epub 2025 Apr 30. Cancer Gene Ther. 2025. PMID: 40307579 Review.
-
Extracellular Vesicles for Clinical Diagnostics: From Bulk Measurements to Single-Vesicle Analysis.ACS Nano. 2025 Aug 12;19(31):28021-28109. doi: 10.1021/acsnano.5c00706. Epub 2025 Jul 28. ACS Nano. 2025. PMID: 40720603 Free PMC article. Review.
-
Implications of Genetic Factors Underlying Mouse Hydronephrosis: Cautionary Considerations on Phenotypic Interpretation in Genetically Engineered Mice.Int J Mol Sci. 2024 Jun 29;25(13):7203. doi: 10.3390/ijms25137203. Int J Mol Sci. 2024. PMID: 39000307 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials

