Isolation and Culture of Primary Fibroblasts from Neonatal Murine Hearts to Study Cardiac Fibrosis
- PMID: 36845532
- PMCID: PMC9947550
- DOI: 10.21769/BioProtoc.4616
Isolation and Culture of Primary Fibroblasts from Neonatal Murine Hearts to Study Cardiac Fibrosis
Abstract
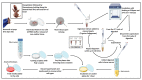
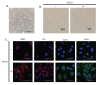
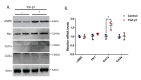
Cardiac fibroblasts are one of the major constituents of a healthy heart. Cultured cardiac fibroblasts are a crucial resource for conducting studies on cardiac fibrosis. The existing methods for culturing cardiac fibroblasts involve complicated steps and require special reagents and instruments. The major problems faced with primary cardiac fibroblast culture are the low yield and viability of the cultured cells and contamination with other heart cell types, including cardiomyocytes, endothelial cells, and immune cells. Numerous parameters, including the quality of the reagents used for the culture, conditions maintained during digestion of the cardiac tissue, composition of the digestion mixture used, and age of the pups used for culture determine the yield and purity of the cultured cardiac fibroblasts. The present study describes a detailed and simplified protocol to isolate and culture primary cardiac fibroblasts from neonatal murine pups. We demonstrate the transdifferentiation of fibroblasts into myofibroblasts through transforming growth factor (TGF)-β1 treatment, representing the changes in fibroblasts during cardiac fibrosis. These cells can be used to study the various aspects of cardiac fibrosis, inflammation, fibroblast proliferation, and growth.
Keywords: Cardiac fibroblasts; Cardiac fibrosis; Fibrosis; Primary culture; TGF-β1.
Copyright © 2023 The Authors; This is an open access article under the CC BY-NC license (https://creativecommons.org/licenses/by-nc/4.0/).
Conflict of interest statement
Competing interestsThe authors declare no conflict of interest. Data Availability Statement The data supporting the study are available to the corresponding author upon request.
Figures
References
-
- Alikhani Z., Alikhani M., Boyd C. M., Nagao K., Trackman P. C. and Graves D. T.(2005). Advanced glycation end products enhance expression of pro-apoptotic genes and stimulate fibroblast apoptosis through cytoplasmic and mitochondrial pathways. J Biol Chem 280(13): 12087-12095. - PubMed
-
- Camelliti P., Borg T. K. and Kohl P.(2005). Structural and functional characterisation of cardiac fibroblasts. Cardiovasc Res 65(1): 40-51. - PubMed
-
- Chacar S., Farès N., Bois P. and Faivre J. F.(2017). Basic Signaling in Cardiac Fibroblasts. J Cell Physiol 232(4): 725-730. - PubMed
-
- desJardins-Park H. E., Foster D. S. and Longaker M. T.(2018). Fibroblasts and wound healing: an update. Regenerative Medicine 13(5): 491-495. - PubMed
-
- Eghbali M.(1992). Cardiac fibroblasts: function, regulation of gene expression, and phenotypic modulation. Basic Res Cardiol 2: 183–189.. - PubMed
LinkOut - more resources
Full Text Sources

