Plasma and Urine Free Glycosaminoglycans as Monitoring and Predictive Biomarkers in Metastatic Renal Cell Carcinoma: A Prospective Cohort Study
- PMID: 36848607
- PMCID: PMC10309541
- DOI: 10.1200/PO.22.00361
Plasma and Urine Free Glycosaminoglycans as Monitoring and Predictive Biomarkers in Metastatic Renal Cell Carcinoma: A Prospective Cohort Study
Abstract
Purpose: No liquid biomarkers are approved in metastatic renal cell carcinoma (mRCC) despite the need to predict and monitor response noninvasively to tailor treatment choices. Urine and plasma free glycosaminoglycan profiles (GAGomes) are promising metabolic biomarkers in mRCC. The objective of this study was to explore if GAGomes could predict and monitor response in mRCC.
Patients and methods: We enrolled a single-center prospective cohort of patients with mRCC elected for first-line therapy (ClinicalTrials.gov identifier: NCT02732665) plus three retrospective cohorts (ClinicalTrials.gov identifiers: NCT00715442 and NCT00126594) for external validation. Response was dichotomized as progressive disease (PD) versus non-PD every 8-12 weeks. GAGomes were measured at treatment start, after 6-8 weeks, and every third month in a blinded laboratory. We correlated GAGomes with response and developed scores to classify PD versus non-PD, which were used to predict response at treatment start or after 6-8 weeks.
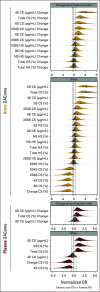
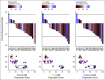
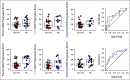
Results: Fifty patients with mRCC were prospectively included, and all received tyrosine kinase inhibitors (TKIs). PD correlated with alterations in 40% of GAGome features. We developed plasma, urine, and combined glycosaminoglycan progression scores that monitored PD at each response evaluation visit with the area under the receiving operating characteristic curve (AUC) of 0.93, 0.97, and 0.98, respectively. For internal validation, the scores predicted PD at treatment start with the AUC of 0.66, 0.68, and 0.74 and after 6-8 weeks with the AUC of 0.76, 0.66, and 0.75. For external validation, 70 patients with mRCC were retrospectively included and all received TKI-containing regimens. The plasma score predicted PD at treatment start with the AUC of 0.90 and at 6-8 weeks with the AUC of 0.89. The pooled sensitivity and specificity were 58% and 79% at treatment start. Limitations include the exploratory study design.
Conclusion: GAGomes changed in association with mRCC response to TKIs and may provide biologic insights into mRCC mechanisms of response.
Conflict of interest statement
The following represents disclosure information provided by authors of this manuscript. All relationships are considered compensated unless otherwise noted. Relationships are self-held unless noted. I = Immediate Family Member, Inst = My Institution. Relationships may not relate to the subject matter of this manuscript. For more information about ASCO's conflict of interest policy, please refer to
Open Payments is a public database containing information reported by companies about payments made to US-licensed physicians (
Figures



References
-
- Moch H, Srigley J, Delahunt B, et al. Biomarkers in renal cancer. Virchows Arch. 2014;464:359–365. - PubMed
-
- Kotecha RR, Motzer RJ, Voss MH. Towards individualized therapy for metastatic renal cell carcinoma. Nat Rev Clin Oncol. 2019;16:621–633. - PubMed
-
- Escudier B, Porta C, Schmidinger M, et al. Renal cell carcinoma: ESMO clinical practice guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2019;30:706–720. - PubMed
-
- Motzer RJ, Jonasch E, Agarwal N, et al. Kidney Cancer, Version 2.2017, NCCN Clinical Practice Guidelines in Oncology J Natl Compr Canc Netw 15(6)804–834.2017 - PubMed
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Medical

