Immunofluorescence assay for demyelination, remyelination, and proliferation in an acute cuprizone mouse model
- PMID: 36853716
- PMCID: PMC9918794
- DOI: 10.1016/j.xpro.2023.102072
Immunofluorescence assay for demyelination, remyelination, and proliferation in an acute cuprizone mouse model
Abstract
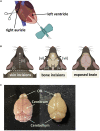
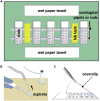
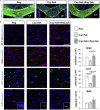
Here, we present a protocol to assess demyelination in the corpus callosum of an acute cuprizone mouse model, which is routinely used to induce demyelination for studying myelin regeneration in the rodent brain. We describe the tracing of neural stem cells via intraperitoneal injection of tamoxifen into adult Gli1CreERT2;Ai9 mice and the induction of demyelination with cuprizone diet. We also detail EdU administration, cryosectioning of the mouse brain, EdU labeling, and immunofluorescence staining to examine proliferation and myelination. For complete details on the use and execution of this protocol, please refer to Radecki et al. (2020).1.
Keywords: Cell Biology; Microscopy; Model Organisms; Molecular/Chemical Probes; Neuroscience; Stem Cells.
Copyright © 2023 The Author(s). Published by Elsevier Inc. All rights reserved.
Conflict of interest statement
Declaration of interests J.S. is a co-inventor in the patent US 9,248,128 and a consultant for GliXogen Therapeutics.
Figures


References
-
- Radecki D.Z., Messling H.M., Haggerty-Skeans J.R., Bhamidipati S.K., Clawson E.D., Overman C.A., Thatcher M.M., Salzer J.L., Samanta J. Relative levels of Gli1 and Gli2 determine the response of ventral neural stem cells to demyelination. Stem Cell Rep. 2020;15:1047–1055. doi: 10.1016/j.stemcr.2020.10.003. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

