Combining radiation and the ATR inhibitor berzosertib activates STING signaling and enhances immunotherapy via inhibiting SHP1 function in colorectal cancer
- PMID: 36855844
- PMCID: PMC10091106
- DOI: 10.1002/cac2.12412
Combining radiation and the ATR inhibitor berzosertib activates STING signaling and enhances immunotherapy via inhibiting SHP1 function in colorectal cancer
Abstract
Background: Immune checkpoint inhibitors (ICIs) targeting programmed cell death protein 1 (PD-1) and programmed death-ligand 1 (PD-L1) have shown a moderate response in colorectal cancer (CRC) with deficient mismatch repair (dMMR) functions and poor response in patients with proficient MMR (pMMR). pMMR tumors are generally immunogenically "cold", emphasizing combination strategies to turn the "cold" tumor "hot" to enhance the efficacy of ICIs. ATR inhibitors (ATRi) have been proven to cooperate with radiation to promote antitumor immunity, but it is unclear whether ATRi could facilitate the efficacy of IR and ICI combinations in CRCs. This study aimed to investigate the efficacy of combining ATRi, irradiation (IR), and anti-PD-L1 antibodies in CRC mouse models with different microsatellite statuses.
Methods: The efficacy of combining ATRi, IR, and anti-PD-L1 antibodies was evaluated in CRC tumors. The tumor microenvironment and transcriptome signatures were investigated under different treatment regimens. The mechanisms were explored via cell viability assay, flow cytometry, immunofluorescence, immunoblotting, co-immunoprecipitation, and real-time quantitative PCR in multiple murine and human CRC cell lines.
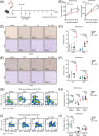
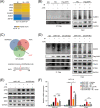
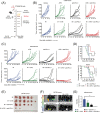
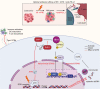
Results: Combining ATRi berzosertib and IR enhanced CD8+ T cell infiltration and enhanced the efficacy of anti-PD-L1 therapy in mouse CRC models with different microsatellite statuses. The mechanistic study demonstrated that IR + ATRi could activate both the canonical cGAS-STING-pTBK1/pIRF3 axis by increasing cytosolic double-stranded DNA levels and the non-canonical STING signaling by attenuating SHP1-mediated inhibition of the TRAF6-STING-p65 axis, via promoting SUMOylation of SHP1 at lysine 127. By boosting the STING signaling, IR + ATRi induced type I interferon-related gene expression and strong innate immune activation and reinvigorated the cold tumor microenvironment, thus facilitating immunotherapy.
Conclusions: The combination of ATRi and IR could facilitate anti-PD-L1 therapy by promoting STING signaling in CRC models with different microsatellite statuses. The new combination strategy raised by our study is worth investigating in the management of CRC.
Keywords: ATR inhibitor; DNA damage; PD-L1; SHP1; SUMOylation; cGAS-STING; colorectal cancer; immune checkpoint inhibitor; innate immunity; radiotherapy.
© 2023 The Authors. Cancer Communications published by John Wiley & Sons Australia, Ltd. on behalf of Sun Yat-sen University Cancer Center.
Conflict of interest statement
All authors have no conflicts of interests to declare.
Figures



References
-
- Siegel RL, Miller KD, Jemal A. Cancer statistics, 2020. CA: A Cancer Journal for Clinicians. 2020;70(1):7‐30. - PubMed
-
- van der Geest LGM, Jt Lam‐Boer, Koopman M, Verhoef C, Elferink MAG, de Wilt JHW. Nationwide trends in incidence, treatment and survival of colorectal cancer patients with synchronous metastases. Clinical & experimental metastasis. 2015;32(5):457‐65. - PubMed
-
- Killock D. Immunotherapy: PD‐1 blockade exploiting MMR deficiency. Nature Reviews Clinical Oncology. 2017;14(8):459. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

