Nephroprotective Effects of Dapagliflozin in Patients with Type 2 Diabetes
- PMID: 36858619
- PMCID: PMC10037009
- DOI: 10.2169/internalmedicine.6685-20
Nephroprotective Effects of Dapagliflozin in Patients with Type 2 Diabetes
Abstract
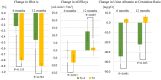
Objective This study analyzed changes in the estimated glomerular filtration rate calculated using cystatin C (eGFRcys) and sodium excretion in the urine after administering dapagliflozin as an add-on therapy to conventional treatment for diabetes. Methods This was a single-center, single-group, prospective interventional study. Dapagliflozin was administered to improve the plasma glucose control in 30 subjects with type 2 diabetes mellitus (age 53±8 years old; 66.6% men). Blood and urine tests were performed before and 6 and 12 months after dapagliflozin administration. The daily sodium excretion was estimated with the Kawasaki formula using second-morning urine samples. Results The eGFRcys did not markedly differ before and 6 months after the dapagliflozin administration but was significantly increased after 12 months (p<0.001), and the estimated daily sodium excretion was also significantly increased (p<0.001 at 6 months and p=0.002 at 12 months). The systolic and diastolic blood pressures tended to decrease after administration. The HbA1c level after the administration of dapagliflozin tended to be lower in the T3 group, showing the smallest increase in changes in the estimated daily sodium excretion from baseline to 6 months (28.2-107.5 mEq/day), than in the combined groups of T1 (219.5-110.1 mEq/day) and T2 (101.4-28.9 mEq/day). In contrast, the eGFRcys was significantly higher in the combined groups of T1 and T2 than that in the T3 group at both 6 and 12 months (p=0.031 and p=0.007, respectively). Conclusions Add-on therapy with dapagliflozin increased the urinary sodium excretion and decreased the blood pressure even in the early phase of this therapy. Our results suggest that dapagliflozin add-on therapy may exert nephroprotective effects in subjects with type 2 diabetes mellitus.
Keywords: dapagliflozin; diabetes mellitus; nephroprotective effects; sodium excretion; sodium glucose cotransporter-2.
Conflict of interest statement
Masato Odawara: Honoraria, Daiichi Sankyo, MSD, Ono, Novartis, Astellas, Sanwa Kagaku Kenkyusho, AstraZeneca, Kyowa Hakko Kirin, Kowa, Takeda, Tanabe Mitsubishi, Eli Lilly, Nippon Boehringer, Sanofi, Novo Nordisk, Dainippon Sumitomo and Taisho Toyama; Research funding, Dainippon Sumitomo, Eli Lilly, Daiichi Sankyo, Ono, Astellas, Kyowa Hakko Kirin, Takeda, Tanabe Mitsubishi and Nippon Boehringer.
Figures

References
-
- Cheung N, Wang JJ, Klein R, et al. . Diabetic retinopathy and the risk of coronary heart disease: the atherosclerosis risk in communities study. Diabetes Care 30: 1742-1746, 2007. - PubMed
-
- Patel A, MacMahon S, Chalmers J, et al. . Intensive plasma glucose control and vascular outcomes in subjects with type 2 diabetes. N Engl J Med 358: 2560-2572, 2008. - PubMed
-
- Papademetriou V, Lovato L, Doumas M, et al. . Chronic kidney disease and intensive glycemic control increase cardiovascular risk in subjects with type 2 diabetes. Kidney Int 87: 649-659, 2015. - PubMed

