Short-term tamoxifen administration improves hepatic steatosis and glucose intolerance through JNK/MAPK in mice
- PMID: 36864030
- PMCID: PMC9981902
- DOI: 10.1038/s41392-022-01299-y
Short-term tamoxifen administration improves hepatic steatosis and glucose intolerance through JNK/MAPK in mice
Abstract
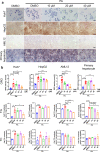
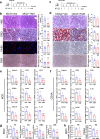
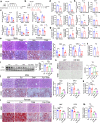
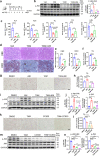
Nonalcoholic fatty liver disease (NAFLD) which is a leading cause of chronic liver diseases lacks effective treatment. Tamoxifen has been proven to be the first-line chemotherapy for several solid tumors in clinics, however, its therapeutic role in NAFLD has never been elucidated before. In vitro experiments, tamoxifen protected hepatocytes against sodium palmitate-induced lipotoxicity. In male and female mice fed with normal diets, continuous tamoxifen administration inhibited lipid accumulation in liver, and improved glucose and insulin intolerance. Short-term tamoxifen administration largely improved hepatic steatosis and insulin resistance, however, the phenotypes manifesting inflammation and fibrosis remained unchanged in abovementioned models. In addition, mRNA expressions of genes related to lipogenesis, inflammation, and fibrosis were downregulated by tamoxifen treatment. Moreover, the therapeutic effect of tamoxifen on NAFLD was not gender or ER dependent, as male and female mice with metabolic disorders shared no difference in response to tamoxifen and ER antagonist (fulvestrant) did not abolish its therapeutic effect as well. Mechanistically, RNA sequence of hepatocytes isolated from fatty liver revealed that JNK/MAPK signaling pathway was inactivated by tamoxifen. Pharmacological JNK activator (anisomycin) partially deprived the therapeutic role of tamoxifen in treating hepatic steatosis, proving tamoxifen improved NAFLD in a JNK/MAPK signaling-dependent manner.
© 2022. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures



References
-
- Ioannou GN. Epidemiology and risk-stratification of NAFLD-associated HCC. J. Hepatol. 2021;S0168-8278:02007–02009. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials

