Delivery challenges for CRISPR-Cas9 genome editing for Duchenne muscular dystrophy
- PMID: 36864908
- PMCID: PMC9969352
- DOI: 10.1063/5.0131452
Delivery challenges for CRISPR-Cas9 genome editing for Duchenne muscular dystrophy
Abstract
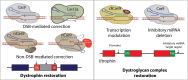
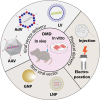
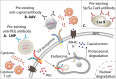
Duchene muscular dystrophy (DMD) is an X-linked neuromuscular disorder that affects about one in every 5000 live male births. DMD is caused by mutations in the gene that codes for dystrophin, which is required for muscle membrane stabilization. The loss of functional dystrophin causes muscle degradation that leads to weakness, loss of ambulation, cardiac and respiratory complications, and eventually, premature death. Therapies to treat DMD have advanced in the past decade, with treatments in clinical trials and four exon-skipping drugs receiving conditional Food and Drug Administration approval. However, to date, no treatment has provided long-term correction. Gene editing has emerged as a promising approach to treating DMD. There is a wide range of tools, including meganucleases, zinc finger nucleases, transcription activator-like effector nucleases, and, most notably, RNA-guided enzymes from the bacterial adaptive immune system clustered regularly interspaced short palindromic repeats (CRISPR). Although challenges in using CRISPR for gene therapy in humans still abound, including safety and efficiency of delivery, the future for CRISPR gene editing for DMD is promising. This review will summarize the progress in CRISPR gene editing for DMD including key summaries of current approaches, delivery methodologies, and the challenges that gene editing still faces as well as prospective solutions.
© 2023 Author(s).
Conflict of interest statement
Christopher E. Nelson has patent applications related to genome editing for DMD.
Figures
Similar articles
-
Restoration of dystrophin expression and correction of Duchenne muscular dystrophy by genome editing.Expert Opin Biol Ther. 2021 Aug;21(8):1049-1061. doi: 10.1080/14712598.2021.1872539. Epub 2021 Jan 25. Expert Opin Biol Ther. 2021. PMID: 33401973 Review.
-
Gene modification strategies using AO-mediated exon skipping and CRISPR/Cas9 as potential therapies for Duchenne muscular dystrophy patients.Eng Biol. 2020 Dec 9;4(3):37-42. doi: 10.1049/enb.2020.0017. eCollection 2020 Dec. Eng Biol. 2020. PMID: 36968157 Free PMC article.
-
CRISPR Therapeutics for Duchenne Muscular Dystrophy.Int J Mol Sci. 2022 Feb 6;23(3):1832. doi: 10.3390/ijms23031832. Int J Mol Sci. 2022. PMID: 35163754 Free PMC article. Review.
-
CRISPR-Based Gene Therapies: From Preclinical to Clinical Treatments.Cells. 2024 May 8;13(10):800. doi: 10.3390/cells13100800. Cells. 2024. PMID: 38786024 Free PMC article. Review.
-
Molecular correction of Duchenne muscular dystrophy by splice modulation and gene editing.RNA Biol. 2021 Jul;18(7):1048-1062. doi: 10.1080/15476286.2021.1874161. Epub 2021 Jan 20. RNA Biol. 2021. PMID: 33472516 Free PMC article. Review.
Cited by
-
Advancing CRISPR genome editing into gene therapy clinical trials: progress and future prospects.Expert Rev Mol Med. 2025 Mar 31;27:e16. doi: 10.1017/erm.2025.10. Expert Rev Mol Med. 2025. PMID: 40160040 Free PMC article. Review.
-
Gene Editing for Duchenne Muscular Dystrophy: From Experimental Models to Emerging Therapies.Degener Neurol Neuromuscul Dis. 2025 Apr 12;15:17-40. doi: 10.2147/DNND.S495536. eCollection 2025. Degener Neurol Neuromuscul Dis. 2025. PMID: 40241992 Free PMC article. Review.
-
Beyond the Cut: Long-read sequencing reveals complex genomic and transcriptomic changes in AAV-CRISPR therapy for Duchenne Muscular Dystrophy.bioRxiv [Preprint]. 2025 Aug 1:2025.08.01.668007. doi: 10.1101/2025.08.01.668007. bioRxiv. 2025. PMID: 40766352 Free PMC article. Preprint.
References
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous
