This is a preprint.
Integrating lipid metabolism, pheromone production and perception by Fruitless and Hepatocyte nuclear factor 4
- PMID: 36865119
- PMCID: PMC9980076
- DOI: 10.1101/2023.02.23.529767
Integrating lipid metabolism, pheromone production and perception by Fruitless and Hepatocyte nuclear factor 4
Update in
-
Integrating lipid metabolism, pheromone production and perception by Fruitless and Hepatocyte Nuclear Factor 4.Sci Adv. 2023 Jun 30;9(26):eadf6254. doi: 10.1126/sciadv.adf6254. Epub 2023 Jun 30. Sci Adv. 2023. PMID: 37390217 Free PMC article.
Abstract
Sexual attraction and perception, governed by separate genetic circuits in different organs, are crucial for mating and reproductive success, yet the mechanisms of how these two aspects are integrated remain unclear. In Drosophila , the male-specific isoform of Fruitless (Fru), Fru M , is known as a master neuro-regulator of innate courtship behavior to control perception of sex pheromones in sensory neurons. Here we show that the non-sex specific Fru isoform (Fru COM ) is necessary for pheromone biosynthesis in hepatocyte-like oenocytes for sexual attraction. Loss of Fru COM in oenocytes resulted in adults with reduced levels of the cuticular hydrocarbons (CHCs), including sex pheromones, and show altered sexual attraction and reduced cuticular hydrophobicity. We further identify Hepatocyte nuclear factor 4 ( Hnf4 ) as a key target of Fru COM in directing fatty acid conversion to hydrocarbons in adult oenocytes. fru - and Hnf4 -depletion disrupts lipid homeostasis, resulting in a novel sex-dimorphic CHC profile, which differs from doublesex - and transformer -dependent sexual dimorphism of the CHC profile. Thus, Fru couples pheromone perception and production in separate organs for precise coordination of chemosensory communication that ensures efficient mating behavior.
Teaser: Fruitless and lipid metabolism regulator HNF4 integrate pheromone biosynthesis and perception to ensure robust courtship behavior.
Conflict of interest statement
Figures

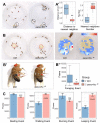
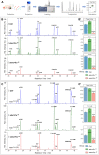

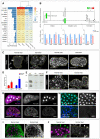
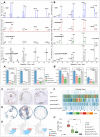


Similar articles
-
Integrating lipid metabolism, pheromone production and perception by Fruitless and Hepatocyte Nuclear Factor 4.Sci Adv. 2023 Jun 30;9(26):eadf6254. doi: 10.1126/sciadv.adf6254. Epub 2023 Jun 30. Sci Adv. 2023. PMID: 37390217 Free PMC article.
-
Chromatin-based reprogramming of a courtship regulator by concurrent pheromone perception and hormone signaling.Sci Adv. 2020 May 22;6(21):eaba6913. doi: 10.1126/sciadv.aba6913. eCollection 2020 May. Sci Adv. 2020. PMID: 32494751 Free PMC article.
-
fruitless is sex-differentially spliced and is important for the courtship behavior and development of silkmoth Bombyx mori.Insect Biochem Mol Biol. 2023 Aug;159:103989. doi: 10.1016/j.ibmb.2023.103989. Epub 2023 Jul 13. Insect Biochem Mol Biol. 2023. PMID: 37453662
-
From fruitless to sex: On the generation and diversification of an innate behavior.Genes Brain Behav. 2021 Nov;20(8):e12772. doi: 10.1111/gbb.12772. Epub 2021 Oct 21. Genes Brain Behav. 2021. PMID: 34672079 Review.
-
Neurogenetics of courtship and mating in Drosophila.Adv Genet. 2008;62:67-184. doi: 10.1016/S0065-2660(08)00603-2. Adv Genet. 2008. PMID: 19010254 Review.
References
-
- Wyatt T. D., Pheromones and signature mixtures: defining species-wide signals and variable cues for identity in both invertebrates and vertebrates. Journal of Comparative Physiology A 196, 685–700 (2010). - PubMed
-
- Toolson E. C., Effects of rearing temperature on cuticle permeability and epicuticular lipid composition in Drosophila pseudoobscura. Journal of Experimental Zoology 222, 249–253 (1982).
-
- Lockey K. H., Lipids of the insect cuticle: origin, composition and function. Comparative Biochemistry and Physiology Part B: Comparative Biochemistry 89, 595–645 (1988).
-
- Rouault J.-D., Marican C., Wicker-Thomas C., Jallon J.-M., in Drosophila melanogaster, Drosophila simulans: So Similar, So Different. (Springer, 2004), pp. 195–212. - PubMed
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
