Heparan sulfates and heparan sulfate binding proteins in sepsis
- PMID: 36865384
- PMCID: PMC9971734
- DOI: 10.3389/fmolb.2023.1146685
Heparan sulfates and heparan sulfate binding proteins in sepsis
Abstract
Heparan sulfates (HSs) are the main components in the glycocalyx which covers endothelial cells and modulates vascular homeostasis through interactions with multiple Heparan sulfate binding proteins (HSBPs). During sepsis, heparanase increases and induces HS shedding. The process causes glycocalyx degradation, exacerbating inflammation and coagulation in sepsis. The circulating heparan sulfate fragments may serve as a host defense system by neutralizing dysregulated Heparan sulfate binding proteins or pro-inflammatory molecules in certain circumstances. Understanding heparan sulfates and heparan sulfate binding proteins in health and sepsis is critical to decipher the dysregulated host response in sepsis and advance drug development. In this review, we will overview the current understanding of HS in glycocalyx under septic condition and the dysfunctional heparan sulfate binding proteins as potential drug targets, particularly, high mobility group box 1 (HMGB1) and histones. Moreover, several drug candidates based on heparan sulfates or related to heparan sulfates, such as heparanase inhibitors or heparin-binding protein (HBP), will be discussed regarding their recent advances. By applying chemical or chemoenzymatic approaches, the structure-function relationship between heparan sulfates and heparan sulfate binding proteins is recently revealed with structurally defined heparan sulfates. Such homogenous heparan sulfates may further facilitate the investigation of the role of heparan sulfates in sepsis and the development of carbohydrate-based therapy.
Keywords: HMGB1 (high mobility group box 1); heparan sulfate; heparan sulfate binding proteins; heparin; histones.
Copyright © 2023 Liao, Liu and Arnold.
Conflict of interest statement
JL is a founder and chief scientific officer for Glycan Therapeutics. KA is a principal scientist at Glycan Therapeutics. The JL lab at UNC has received a gift from Glycan Therapeutics to support research in glycoscience. Y-EL declare no competing interests.
Figures


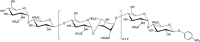


Similar articles
-
Endothelial Heparan Sulfate Mediates Hepatic Neutrophil Trafficking and Injury during Staphylococcus aureus Sepsis.mBio. 2021 Oct 26;12(5):e0118121. doi: 10.1128/mBio.01181-21. Epub 2021 Sep 21. mBio. 2021. PMID: 34544271 Free PMC article.
-
Intravenous fluid resuscitation is associated with septic endothelial glycocalyx degradation.Crit Care. 2019 Jul 23;23(1):259. doi: 10.1186/s13054-019-2534-2. Crit Care. 2019. PMID: 31337421 Free PMC article.
-
Loss of endothelial sulfatase-1 after experimental sepsis attenuates subsequent pulmonary inflammatory responses.Am J Physiol Lung Cell Mol Physiol. 2019 Nov 1;317(5):L667-L677. doi: 10.1152/ajplung.00175.2019. Epub 2019 Aug 28. Am J Physiol Lung Cell Mol Physiol. 2019. PMID: 31461325 Free PMC article.
-
Endothelial Heparan Sulfate Proteoglycans in Sepsis: The Role of the Glycocalyx.Semin Thromb Hemost. 2021 Apr;47(3):274-282. doi: 10.1055/s-0041-1725064. Epub 2021 Apr 1. Semin Thromb Hemost. 2021. PMID: 33794552 Review.
-
Targeting the "sweet spot" in septic shock - A perspective on the endothelial glycocalyx regulating proteins Heparanase-1 and -2.Matrix Biol Plus. 2021 Dec 2;12:100095. doi: 10.1016/j.mbplus.2021.100095. eCollection 2021 Dec. Matrix Biol Plus. 2021. PMID: 34917926 Free PMC article. Review.
Cited by
-
The Effects of Heparan Sulfate Infusion on Endothelial and Organ Injury in a Rat Pneumosepsis Model.J Clin Med. 2023 Oct 10;12(20):6438. doi: 10.3390/jcm12206438. J Clin Med. 2023. PMID: 37892576 Free PMC article.
-
Role of heparanase in pulmonary hypertension.Front Pharmacol. 2023 Aug 11;14:1202676. doi: 10.3389/fphar.2023.1202676. eCollection 2023. Front Pharmacol. 2023. PMID: 37637421 Free PMC article. Review.
-
CIRCULATING HEPARAN SULFATE PROFILES IN PEDIATRIC ACUTE RESPIRATORY DISTRESS SYNDROME.Shock. 2024 Oct 1;62(4):496-504. doi: 10.1097/SHK.0000000000002421. Shock. 2024. PMID: 39331799 Free PMC article.
-
By activating endothelium histone H4 mediates oleic acid-induced acute respiratory distress syndrome.BMC Pulm Med. 2025 Jan 6;25(1):3. doi: 10.1186/s12890-024-03334-w. BMC Pulm Med. 2025. PMID: 39757148 Free PMC article.
-
The endothelial glycocalyx-All the same? No, it is not.Acute Med Surg. 2023 Oct 4;10(1):e896. doi: 10.1002/ams2.896. eCollection 2023 Jan-Dec. Acute Med Surg. 2023. PMID: 37808968 Free PMC article. Review.
References
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

