Adverse events of tumor necrosis factor alpha inhibitors for the treatment of ankylosing spondylitis: A meta-analysis of randomized, placebo-controlled trials
- PMID: 36865909
- PMCID: PMC9972296
- DOI: 10.3389/fphar.2023.1084614
Adverse events of tumor necrosis factor alpha inhibitors for the treatment of ankylosing spondylitis: A meta-analysis of randomized, placebo-controlled trials
Abstract
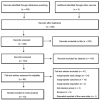
Objective: Tumor necrosis factor alpha inhibitors (TNFi) have shown substantial efficacy in alleviating and treating ankylosing spondylitis (AS). However, the heightened interest is accompanied by concerns over adverse events. In this meta-analysis, we analyzed both serious and common adverse events in patients treated with tumor necrosis factor alpha inhibitors compared with those in the placebo group. Methods: We searched for clinical trials in PubMed, Embase, Cochrane Library, China National Knowledge Infrastructure, Wanfang Data, and VIP Data. Studies were selected based on strict inclusion and exclusion criteria. Only randomized, placebo-controlled trials were included in the final analysis. RevMan 5.4 software was used for performing meta-analyses. Results: A total of 18 randomized controlled trials recruiting 3,564 patients with ankylosing spondylitis were included, with overall moderate to high methodological quality. Compared with the placebo group, the incidences showed no difference and were only slightly increased numerically for serious adverse events, serious infections, upper respiratory tract infection, and malignancies in patients treated with tumor necrosis factor alpha inhibitors. However, tumor necrosis factor alpha inhibitor treatment significantly increased the incidence of overall adverse events, nasopharyngitis, headache, and injection-site reactions in ankylosing spondylitis patients when compared with placebo. Conclusion: The available data indicated that ankylosing spondylitis patients who received tumor necrosis factor alpha inhibitors had no significantly increased risks of serious adverse events when compared with the placebo group. However, tumor necrosis factor alpha inhibitors significantly increased the incidence rate of common adverse events, including nasopharyngitis, headache, and injection-site reactions. Large-scale and long-term follow-up clinical trials are still necessary to further investigate the safety of tumor necrosis factor alpha inhibitors in ankylosing spondylitis treatment.
Keywords: adverse events; ankylosing spondylitis; infection; randomized controlled trial; tumor necrosis factor alpha inhibitors.
Copyright © 2023 Feng, Zhao, Kuang, Dai, Cen and Qin.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures




Similar articles
-
The Comparative Safety of TNF Inhibitors in Ankylosing Spondylitis-a Meta-Analysis Update of 14 Randomized Controlled Trials.Clin Rev Allergy Immunol. 2018 Apr;54(2):234-243. doi: 10.1007/s12016-017-8623-6. Clin Rev Allergy Immunol. 2018. PMID: 28717941
-
Golimumab: Review of the efficacy and tolerability of a recently approved tumor necrosis factor-α inhibitor.Clin Ther. 2010 Sep;32(10):1681-703. doi: 10.1016/j.clinthera.2010.09.003. Clin Ther. 2010. PMID: 21194591 Review.
-
Effect of TNF-inhibitor therapy on spinal structural progression in ankylosing spondylitis patients: A systematic review and meta-analysis.Int J Rheum Dis. 2020 Jun;23(6):728-743. doi: 10.1111/1756-185X.13829. Epub 2020 May 17. Int J Rheum Dis. 2020. PMID: 32419337
-
The Safety Profile of Tumor Necrosis Factor Inhibitors in Ankylosing Spondylitis: Are TNF Inhibitors Safer Than We Thought?J Clin Pharmacol. 2019 Apr;59(4):445-462. doi: 10.1002/jcph.1348. Epub 2018 Nov 26. J Clin Pharmacol. 2019. PMID: 30476367 Review.
-
Efficacy and safety of filgotinib, a selective Janus kinase 1 inhibitor, in patients with active ankylosing spondylitis (TORTUGA): results from a randomised, placebo-controlled, phase 2 trial.Lancet. 2018 Dec 1;392(10162):2378-2387. doi: 10.1016/S0140-6736(18)32463-2. Epub 2018 Oct 22. Lancet. 2018. PMID: 30360970 Clinical Trial.
Cited by
-
Treatment patterns and factors associated with discontinuation of monoclonal antibodies.SAGE Open Med. 2024 Aug 19;12:20503121241271817. doi: 10.1177/20503121241271817. eCollection 2024. SAGE Open Med. 2024. PMID: 39165864 Free PMC article.
-
Proteome-wide Mendelian randomization identifies therapeutic targets for ankylosing spondylitis.Front Immunol. 2024 Mar 19;15:1366736. doi: 10.3389/fimmu.2024.1366736. eCollection 2024. Front Immunol. 2024. PMID: 38566994 Free PMC article.
-
A full-spectrum Boswellia serrata extract with enhanced bioavailability, and its co-delivered system with curcumin alleviate pain and stiffness associated with moderate spondylitis: a randomized double-blind, placebo-controlled, 3-arm study.Front Pharmacol. 2025 Jul 1;16:1577429. doi: 10.3389/fphar.2025.1577429. eCollection 2025. Front Pharmacol. 2025. PMID: 40667507 Free PMC article.
-
Biomarkers for successful tapering of a tumor necrosis factor inhibitor in patients with radiographic axial spondyloarthritis: A pilot study.SAGE Open Med. 2025 Apr 12;13:20503121251330812. doi: 10.1177/20503121251330812. eCollection 2025. SAGE Open Med. 2025. PMID: 40291149 Free PMC article.
-
A systematic review of tumor necrosis factor-α blockers, anti-interleukins, and small molecule inhibitors for dissecting cellulitis of the scalp treatment.Orphanet J Rare Dis. 2025 May 18;20(1):236. doi: 10.1186/s13023-025-03720-5. Orphanet J Rare Dis. 2025. PMID: 40383754 Free PMC article.
References
-
- Bao C., Huang F., Khan M. A., Fei K., Wu Z., Han C., et al. (2014). Safety and efficacy of golimumab in Chinese patients with active ankylosing spondylitis: 1-year results of a multicentre, randomized, double-blind, placebo-controlled phase III trial. Rheumatol. Oxf. 53, 1654–1663. 10.1093/rheumatology/keu132 - DOI - PubMed
-
- Bongartz T., Sutton A. J., Sweeting M. J., Buchan I., Matteson E. L., Montori V. (2006). Anti-TNF antibody therapy in rheumatoid arthritis and the risk of serious infections and malignancies: Systematic review and meta-analysis of rare harmful effects in randomized controlled trials. JAMA 295, 2275–2285. 10.1001/jama.295.19.2275 - DOI - PubMed
-
- Burmester G. R., Panaccione R., Gordon K. B., McIlraith M. J., Lacerda A. P. (2013). Adalimumab: Long-term safety in 23 458 patients from global clinical trials in rheumatoid arthritis, juvenile idiopathic arthritis, ankylosing spondylitis, psoriatic arthritis, psoriasis and Crohn's disease. Ann. Rheum. Dis. 72, 517–524. 10.1136/annrheumdis-2011-201244 - DOI - PMC - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

