Elucidating the life cycle of opossum parasites: DNA sequences reveal the involvement of planorbid snails as intermediate hosts of Rhopalias spp. (Trematoda: Echinostomatidae) in Brazil
- PMID: 36867609
- PMCID: PMC9983843
- DOI: 10.1371/journal.pone.0279268
Elucidating the life cycle of opossum parasites: DNA sequences reveal the involvement of planorbid snails as intermediate hosts of Rhopalias spp. (Trematoda: Echinostomatidae) in Brazil
Abstract
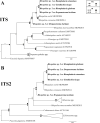
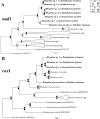
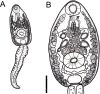
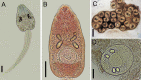
Echinostomatid digeneans belonging to the genus Rhopalias are intestinal trematodes found mainly in opossums in the New World. The genus comprises seven species, but their life cycles and intermediate hosts have been unknown until now. During our long-term study carried out in freshwater habitats within the state of Minas Gerais, Southeast Brazil, echinostomatid cercariae lacking collar spines were found in planorbid snails Biomphalaria glabrata, Biomphalaria straminea, Drepanotrema lucidum and Gundlachia ticaga in six different batches of snail samples collected between 2010 and 2019. Morphologically, the larvae reported herein are morphologically consistent with each other and characterized by the presence of 2-3 large ovoid or spherical corpuscles in each main duct of the excretory system, resembling to Cercaria macrogranulosa previously described from the same region of Brazil. Partial sequences of the ITS (ITS1-5.8S-ITS2) region and 28S gene of the nuclear ribosomal RNA operon, and partial sequences of mitochondrial nad1 and cox1 genes were obtained and compared with the data available for members of the family Echinostomatidae. Nuclear markers indicate that all samples of cercariae evaluated in the present study can be assigned to Rhopalias, but distinct from North American isolates of Rhopalias macracanthus, Rhopalias coronatus and Rhopalias oochi (divergence 0.2-1.2% in 28S and 0.8-4.7% in ITS). The lack of differences verified in both 28S and ITS in 5 out 6 studied samples suggested that they belong to the same species. However, nad1 sequences revealed that our cercariae correspond to three distinct species of Rhopalias (interspecific divergence: 7.7-9.9%), named here as Rhopalias sp. 1, found in B. straminea and G. ticaga, Rhopalias sp. 2 found in B. glabrata and D. lucidum, and Rhopalias sp. 3 also found in D. lucidum. They also differ by 10.8-17.2% from a North American isolate of R. macracanthus sequenced in this study. The cox1 sequences obtained for Rhopalias sp. 1 and Rhopalias sp. 2 (but not Rhopalias sp. 3) reveal that they are distinct from North American isolates of R. macracanthus (genetic divergence 16.3-16.5% and 15.6-15.7%, respectively), R. coronatus (9.2-9.3% and 9.3-9.5%) and Rhopalias oochi (9.0% and 9.5-10.1%). Encysted metacercariae with general morphology similar to that of the body of cercariae were found in tadpoles of Rhinella sp. from the same stream where snails harbored Rhopalias sp. 2, suggesting that the amphibians could act as second intermediate hosts of species of Rhopalias. Data obtained provide the first insights into the life cycle of this unusual echinostomatid genus.
Copyright: © 2023 López-Hernández et al. This is an open access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures


Similar articles
-
Biomphalaria straminea (Mollusca: Planorbidae) as an intermediate host of Drepanocephalus spp. (Trematoda: Echinostomatidae) in Brazil: a morphological and molecular study.Parasitol Res. 2016 Jan;115(1):51-62. doi: 10.1007/s00436-015-4469-0. Epub 2015 May 19. Parasitol Res. 2016. PMID: 25982569
-
Molecular, morphological and experimental-infection studies of cercariae of five species in the superfamily Diplostomoidea (Trematoda: Digenea) infecting Biomphalaria straminea (Mollusca: Planorbidae) in Brazil.Acta Trop. 2019 Nov;199:105082. doi: 10.1016/j.actatropica.2019.105082. Epub 2019 Jul 24. Acta Trop. 2019. PMID: 31351070
-
A putative new genus of avian schistosome transmitted by Biomphalaria straminea (Gastropoda: Planorbidae) in Brazil, with a discussion on the potential involvement in human cercarial dermatitis.Parasitol Int. 2022 Oct;90:102607. doi: 10.1016/j.parint.2022.102607. Epub 2022 Jun 2. Parasitol Int. 2022. PMID: 35659634
-
Prey-mimetism in cercariae of Apatemon (Digenea, Strigeidae) in freshwater in northern latitudes.Parasitol Res. 2023 Mar;122(3):815-831. doi: 10.1007/s00436-023-07779-6. Epub 2023 Jan 21. Parasitol Res. 2023. PMID: 36670312 Review.
-
[Role of gastropods in epidemiology of human parasitic diseases].Wiad Parazytol. 2001;47(1):3-24. Wiad Parazytol. 2001. PMID: 16888946 Review. Polish.
Cited by
-
Parasite-Driven host manipulation: The case of trematodes in Neotropical tadpoles.Int J Parasitol Parasites Wildl. 2024 Nov 26;25:101023. doi: 10.1016/j.ijppaw.2024.101023. eCollection 2024 Dec. Int J Parasitol Parasites Wildl. 2024. PMID: 39687764 Free PMC article.
-
Helminths assemblage in two opossum's species, Didelphis albiventris and Didelphis aurita (Mammalia: Didelphimorphia), from the Atlantic Forest of Argentina.Parasitology. 2025 Apr;152(5):551-562. doi: 10.1017/S0031182025000563. Parasitology. 2025. PMID: 40269638 Free PMC article.
References
-
- Yamaguti S. Synopsis of Digenetic Trematodes of Vertebrates. vol. 1. Keigaku Publishing Company: Tokyo; 1971.
-
- Marshall ME, Miller GC. Some digenetic trematodes from Ecuadorian bats including five new species and one new genus. J. Parasitol. 1979;65: 909–917.
-
- Radev V, Gardner SL, Kanev I. Family Rhopaliidae Looss, 1899. In: Keys to the Trematoda, Jones A, Bray RA, and Gibson DI (eds). London, U.K.: CAB International and The Natural History Museum; 2005. p. 119–120.
-
- Haverkost TR, Gardner SL. A review of species in the genus Rhopalias (Rudolphi, 1819). J. Parasitol. 2008;94: 716–726. - PubMed
MeSH terms
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

