Antagonism of TRPV4 channels partially reduces mechanotransduction in rat skeletal muscle afferents
- PMID: 36869605
- PMCID: PMC10106437
- DOI: 10.1113/JP284026
Antagonism of TRPV4 channels partially reduces mechanotransduction in rat skeletal muscle afferents
Abstract
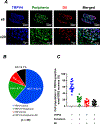
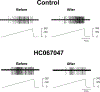
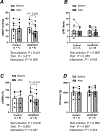
Mechanical distortion of working skeletal muscle induces sympathoexcitation via thin fibre afferents, a reflex response known as the skeletal muscle mechanoreflex. However, to date, the receptor ion channels responsible for mechanotransduction in skeletal muscle remain largely undetermined. Transient receptor potential vanilloid 4 (TRPV4) is known to sense mechanical stimuli such as shear stress or osmotic pressure in various organs. It is hypothesized that TRPV4 in thin-fibre primary afferents innervating skeletal muscle is involved in mechanotransduction. Fluorescence immunostaining revealed that 20.1 ± 10.1% of TRPV4 positive neurons were small dorsal root ganglion (DRG) neurons that were DiI-labelled, and among them 9.5 ± 6.1% of TRPV4 co-localized with the C-fibre marker peripherin. In vitro whole-cell patch clamp recordings from cultured rat DRG neurons demonstrated that mechanically activated current amplitude was significantly attenuated after the application of the TRPV4 antagonist HC067047 compared to control (P = 0.004). Such reductions were also observed in single-fibre recordings from a muscle-nerve ex vivo preparation where HC067047 significantly decreased afferent discharge to mechanical stimulation (P = 0.007). Likewise, in an in vivo decerebrate rat preparation, the renal sympathetic nerve activity (RSNA) and mean arterial pressure (MAP) responses to passive stretch of hindlimb muscle were significantly reduced by intra-arterial injection of HC067047 (ΔRSNA: P = 0.019, ΔMAP: P = 0.002). The findings suggest that TRPV4 plays an important role in mechanotransduction contributing to the cardiovascular responses evoked by the skeletal muscle mechanoreflex during exercise. KEY POINTS: Although a mechanical stimulus to skeletal muscle reflexively activates the sympathetic nervous system, the receptors responsible for mechanotransduction in skeletal muscle thin fibre afferents have not been fully identified. Evidence suggests that TRPV4 is a mechanosensitive channel that plays an important role in mechanotransduction within various organs. Immunocytochemical staining demonstrates that TRPV4 is expressed in group IV skeletal muscle afferents. In addition, we show that the TRPV4 antagonist HC067047 decreases the responsiveness of thin fibre afferents to mechanical stimulation at the muscle tissue level as well as at the level of dorsal root ganglion neurons. Moreover, we demonstrate that intra-arterial HC067047 injection attenuates the sympathetic and pressor responses to passive muscle stretch in decerebrate rats. These data suggest that antagonism of TRPV4 attenuates mechanotransduction in skeletal muscle afferents. The present study demonstrates a probable physiological role for TRPV4 in the regulation of mechanical sensation in somatosensory thin fibre muscle afferents.
Keywords: group IV muscle afferents; mechanotransduction; muscle mechanoreflex; primary sensory neuron; transient receptor potential vanilloid 4.
© 2023 The Authors. The Journal of Physiology published by John Wiley & Sons Ltd on behalf of The Physiological Society.
Conflict of interest statement
COMPETING INTERESTS
The authors declare that they have no competing interests.
Figures


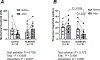

References
-
- Ariano MA, Armstrong RB & Edgerton VR. (1973). Hindlimb muscle fiber populations of five mammals. J Histochem Cytochem 21, 51–55. - PubMed
-
- Bernardi MM & Neto JP. (1979). Effects of abrupt and gradual withdrawal from long-term haloperidol treatment on open field behavior of rats. Psychopharmacology (Berl) 65, 247–250. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

