Paraoxonase 1 and atherosclerosis
- PMID: 36873390
- PMCID: PMC9977831
- DOI: 10.3389/fcvm.2023.1065967
Paraoxonase 1 and atherosclerosis
Abstract
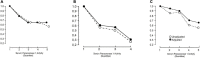
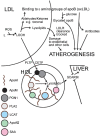
Paraoxonase 1 (PON1), residing almost exclusively on HDL, was discovered because of its hydrolytic activity towards organophosphates. Subsequently, it was also found to hydrolyse a wide range of substrates, including lactones and lipid hydroperoxides. PON1 is critical for the capacity of HDL to protect LDL and outer cell membranes against harmful oxidative modification, but this activity depends on its location within the hydrophobic lipid domains of HDL. It does not prevent conjugated diene formation, but directs lipid peroxidation products derived from these to become harmless carboxylic acids rather than aldehydes which might adduct to apolipoprotein B. Serum PON1 is inversely related to the incidence of new atherosclerotic cardiovascular disease (ASCVD) events, particularly in diabetes and established ASCVD. Its serum activity is frequently discordant with that of HDL cholesterol. PON1 activity is diminished in dyslipidaemia, diabetes, and inflammatory disease. Polymorphisms, most notably Q192R, can affect activity towards some substrates, but not towards phenyl acetate. Gene ablation or over-expression of human PON1 in rodent models is associated with increased and decreased atherosclerosis susceptibility respectively. PON1 antioxidant activity is enhanced by apolipoprotein AI and lecithin:cholesterol acyl transferase and diminished by apolipoprotein AII, serum amyloid A, and myeloperoxidase. PON1 loses this activity when separated from its lipid environment. Information about its structure has been obtained from water soluble mutants created by directed evolution. Such recombinant PON1 may, however, lose the capacity to hydrolyse non-polar substrates. Whilst nutrition and pre-existing lipid modifying drugs can influence PON1 activity there is a cogent need for more specific PON1-raising medication to be developed.
Keywords: PON1 polymorphism; cardiovascular disease; high density lipoprotein; lipid peroxidation; paraoxonase 1; paraoxonase 1 activity.
Copyright © 2023 Durrington, Bashir and Soran.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures


References
-
- Furlong C. Paraoxonase: an historical perspective. In: Ridley A, Frampton J. editors. The paraoxonases: their role in disease development and xenobiotic metabolism. Dordrecht: Springer; (2008).
Publication types
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

