Analyzing the interactome of human CK2β in prostate carcinoma cells reveals HSP70-1 and Rho guanin nucleotide exchange factor 12 as novel interaction partners
- PMID: 36876296
- PMCID: PMC9983076
- DOI: 10.1096/fba.2022-00098
Analyzing the interactome of human CK2β in prostate carcinoma cells reveals HSP70-1 and Rho guanin nucleotide exchange factor 12 as novel interaction partners
Abstract
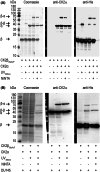
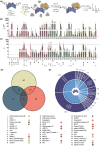
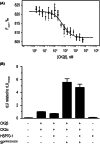
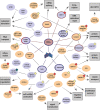
CK2β is the non-catalytic modulating part of the S/T-protein kinase CK2. However, the overall function of CK2β is poorly understood. Here, we report on the identification of 38 new interaction partners of the human CK2β from lysates of DU145 prostate cancer cells using photo-crosslinking and mass spectrometry, whereby HSP70-1 was identified with high abundance. The KD value of its interaction with CK2β was determined as 0.57 μM by microscale thermophoresis, this being the first time, to our knowledge, that a KD value of CK2β with another protein than CK2α or CK2α' was quantified. Phosphorylation studies excluded HSP70-1 as a substrate or activity modulator of CK2, suggesting a CK2 activity independent interaction of HSP70-1 with CK2β. Co-immunoprecipitation experiments in three different cancer cell lines confirmed the interaction of HSP70-1 with CK2β in vivo. A second identified CK2β interaction partner was Rho guanin nucleotide exchange factor 12, indicating an involvement of CK2β in the Rho-GTPase signal pathway, described here for the first time to our knowledge. This points to a role of CK2β in the interaction network affecting the organization of the cytoskeleton.
Keywords: HSP70‐1; Rho guanin nucleotide exchange factor 12; photo‐crosslinking mass spectrometry; prostate cancer; protein kinase CK2.
© 2023 The Authors. FASEB BioAdvances published by Wiley Periodicals LLC on behalf of The Federation of American Societies for Experimental Biology.
Figures



Similar articles
-
Structure of the human protein kinase CK2 catalytic subunit CK2α' and interaction thermodynamics with the regulatory subunit CK2β.J Mol Biol. 2011 Mar 18;407(1):1-12. doi: 10.1016/j.jmb.2011.01.020. Epub 2011 Jan 15. J Mol Biol. 2011. PMID: 21241709
-
The multiple personalities of the regulatory subunit of protein kinase CK2: CK2 dependent and CK2 independent roles reveal a secret identity for CK2beta.Int J Biol Sci. 2005;1(2):67-79. doi: 10.7150/ijbs.1.67. Epub 2005 Apr 1. Int J Biol Sci. 2005. PMID: 15951851 Free PMC article.
-
The regulatory beta subunit of protein kinase CK2 mediates formation of tetrameric CK2 complexes.J Biol Chem. 2000 Feb 18;275(7):5003-10. doi: 10.1074/jbc.275.7.5003. J Biol Chem. 2000. PMID: 10671540
-
Protein kinase CK2 in breast cancer: the CK2β regulatory subunit takes center stage in epithelial plasticity.Cell Mol Life Sci. 2015 Sep;72(17):3305-22. doi: 10.1007/s00018-015-1929-8. Epub 2015 May 20. Cell Mol Life Sci. 2015. PMID: 25990538 Free PMC article. Review.
-
Ecto-protein kinase CK2, the neglected form of CK2.Biomed Rep. 2018 Apr;8(4):307-313. doi: 10.3892/br.2018.1069. Epub 2018 Feb 21. Biomed Rep. 2018. PMID: 29556379 Free PMC article. Review.
Cited by
-
Protein Kinase CK2α', More than a Backup of CK2α.Cells. 2023 Dec 14;12(24):2834. doi: 10.3390/cells12242834. Cells. 2023. PMID: 38132153 Free PMC article. Review.
-
CK2B is a Prognostic Biomarker and a Potential Drug Target for Hepatocellular Carcinoma.Recent Pat Anticancer Drug Discov. 2024;19(5):622-634. doi: 10.2174/0115748928262221230925090120. Recent Pat Anticancer Drug Discov. 2024. PMID: 37779404
References
-
- Heller‐Harrison RA, Meisner H, Czech MP. Cloning and characterization of a cDNA encoding the beta subunit of human casein kinase II. Biochemistry. 1989;28:9053‐9058. - PubMed
-
- Jakobi R, Voss H, Pyerin W. Human phosvitin/casein kinase type II. Molecular cloning and sequencing of full‐length cDNA encoding subunit beta. Eur J Biochem. 1989;183:227‐233. - PubMed
-
- Boldyreff B, Piontek K, Schmidt‐Spaniol I, Issinger OG. The beta subunit of casein kinase II: cloning of cDNAs from murine and porcine origin and expression of the porcine sequence as a fusion protein. Biochim Biophys Acta. 1991;1088:439‐441. - PubMed
-
- Egorova KS, Olenkina OM, Kibanov MV, Kalmykova AI, Gvozdev VA, Olenina LV. Genetically derepressed nucleoplasmic stellate protein in spermatocytes of D. melanogaster interacts with the catalytic subunit of protein kinase 2 and carries histone‐like lysine‐methylated mark. J Mol Biol. 2009;389:895‐906. - PubMed
LinkOut - more resources
Full Text Sources
