Neutrophil extracellular trap formation in anti-neutrophil cytoplasmic antibody-associated and large-vessel vasculitis
- PMID: 36878421
- PMCID: PMC10066833
- DOI: 10.1016/j.clim.2023.109274
Neutrophil extracellular trap formation in anti-neutrophil cytoplasmic antibody-associated and large-vessel vasculitis
Abstract
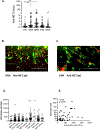
Levels of neutrophil extracellular traps (NETs) were measured in plasma of healthy controls (HC, n = 30) and patients with granulomatosis with polyangiitis (GPA, n = 123), microscopic polyangiitis (MPA, n = 61), Takayasu's arteritis (TAK, n = 58), and giant cell arteritis (GCA, n = 68), at times of remission or activity and correlated with levels of the platelet-derived thrombospondin-1 (TSP-1). Levels of NETs were elevated during active disease in patients with GPA (p < 0.0001), MPA (p = 0.0038), TAK (p < 0.0001), and GCA (p < 0.0001), and in remission for GPA, p < 0.0001, MPA, p = 0.005, TAK, p = 0.03, and GCA, p = 0.0009. All cohorts demonstrated impaired NET degradation. Patients with GPA (p = 0.0045) and MPA (p = 0.005) had anti-NET IgG antibodies. Patients with TAK had anti-histone antibodies (p < 0.01), correlating with presence of NETs. Levels of TSP-1 were increased in all patients with vasculitis, and associated with NET formation. NET formation is a common process in vasculitides. Targeting NET formation or degradation could be potential therapeutic approaches for vasculitides.
Keywords: Anti-neutrophil cytoplasmic antibody associated vasculitis; Histone; Large vessel vasculitis; Neutrophil extracellular traps; Neutrophils; Platelet activation; Thrombospondin-1.
Copyright © 2023 Elsevier Inc. All rights reserved.
Conflict of interest statement
Declaration of Competing Interest Dr. Michailidou received Advisory Board fees from ChemoCentryx. Dr. Khalidi received clinical trial support from BMS, Sanofi and Abbvie, travel support from Astra Zeneca, and Advisory Board fee from Roche. Dr. Koening served on the advisory board for Chemocentryx and Genentech. Dr. Specks reports receiving funds for the following activities: Consulting: AstraZeneca, ChemoCentryx. Research Support: AstraZeneca, GlaxoSmithKline, Bristol-Myers Squibb, Genentech/Roche, InflaRx. Dr. Sreih works at Bristol-Myers Squibb and owns Astra Zeneca and Alexion Stocks. Dr. Warrington received clinical trial support from Eli Lilly and Kiniksa. Dr. Monach received consulting fees from Kiniksa, Celgene/BMC, and ChemoCentryx. Dr. Merkel reports receiving funds for the following activities: Consulting and Research Support: AbbVie, AstraZeneca, Boeringher-Ingelheim, Bristol-Myers Squibb, ChemoCentryx, Forbius, Genentech/Roche, Genzyme/Sanofi, GlaxoSmithKline, InflaRx, Takeda. Consulting only: CSL Behring, Dynacure, EMDSerono, Janssen, Kiniksa, Kyverna, Magenta, MiroBio, Neutrolis, Novartis, Pfizer, Sparrow, Talaris. Royalties: UpToDate. Dr. Lood received research funding from Pfizer, Gilead Sciences, Boehringer Ingelheim, Redd Pharma, Amytryx, and Eli Lilly.
Figures

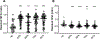

References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

