Interaction of hemorphins with ACE homologs
- PMID: 36878973
- PMCID: PMC9987361
- DOI: 10.1038/s41598-023-30771-0
Interaction of hemorphins with ACE homologs
Abstract
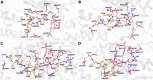
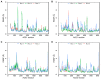
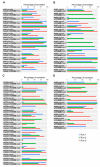
Hemorphins, short bioactive peptides produced by enzymatic cleavage of β-hemoglobin, exhibit antihypertensive properties by inhibiting angiotensin-1 converting enzyme (ACE1). ACE1 is a key player in the renin-angiotensin system (RAS) and regulates blood pressure. ACE1 and its homolog, ACE2, which exhibit opposing activities in the RAS, share considerable similarity in their catalytic domains. The primary objective of this study was to identify and contrast the molecular mechanisms underlying the interaction of hemorphins of camels and that of other mammals with the two ACE homologs. In silico docking and molecular dynamics simulations were performed for ACE1 and ACE2, along with in vitro confirmatory assays for ACE1. The C-domain of ACE1, primarily involved in regulating blood pressure, was used along with the N-terminal peptidase domain of ACE2. The findings revealed conserved hemorphin interactions with equivalent regions of the two ACE homologs and differential residue-level interactions reflecting the substrate preferences of ACE1 and ACE2 considering their opposing functions. Therefore, conserved residue-level associations and implications of poorly conserved regions between the two ACE receptors may potentially guide the discovery of selective domain-specific inhibitors. The findings of this study can provide a basis for the treatment of related disorders in the future.
© 2023. The Author(s).
Conflict of interest statement
R.V. is an editorial board member of Scientific Reports. The other authors declare no competing interests.
Figures




References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

