Medication-induced osteonecrosis of the jaw: a review of cases from the Food and Drug Administration Adverse Event Reporting System (FAERS)
- PMID: 36879299
- PMCID: PMC9987072
- DOI: 10.1186/s40360-023-00657-y
Medication-induced osteonecrosis of the jaw: a review of cases from the Food and Drug Administration Adverse Event Reporting System (FAERS)
Abstract
Background: Osteonecrosis of the jaw (ONJ) is a rare but serious adverse drug reaction (ADR) commonly associated with bisphosphonate and denosumab therapy. Prior research utilized an online, public FDA Adverse Event Reporting System (FAERS) Database to explore this ADR. This data identified and described several novel medications associated with ONJ. Our study aims to build upon the prior findings, reporting trends of medication induced ONJ over time and identifying newly described medications.
Methods: We searched the FAERS database for all reported cases of medication related osteonecrosis of the jaw (MRONJ) from 2010 to 2021. Cases lacking patient age or gender were excluded. Only adults (18 +) and reports from Healthcare Professions were included. Duplicate cases were removed. The top 20 medications were identified and described for April 2010-December 2014 and April 2015-January 2021.
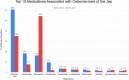
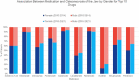
Results: Nineteen thousand six hundred sixty-eight cases of ONJ were reported to the FAERS database from 2010-2021. 8,908 cases met inclusion criteria. 3,132 cases were from 2010-2014 and 5,776 cases from 2015-2021. Within the cases from 2010-2014, 64.7% were female and 35.3% were male, and the average age was 66.1 ± 11.1 years. Between 2015-2021, 64.3% were female and 35.7% were male, and the average age was 69.2 ± 11.5 years. Review of the 2010-2014 data identified several medications and drug classes associated with ONJ not previously described. They include lenalidomide, corticosteroids (prednisolone and dexamethasone), docetaxel and paclitaxel, letrozole, methotrexate, imatinib, and teriparatide. Novel drugs and classes described between 2015-2021 include palbociclib, pomalidomide, radium 223, nivolumab, and cabozantinib.
Discussion: While stricter inclusion criteria and removal of duplicate cases led to fewer overall identified cases of MRONJ when compared to prior research, our data represents a more reliable analysis of MRONJ reports to the FAERS database. Denosumab was the most frequently reported medication associated with ONJ. While unable to imply incidence rates from our data due to the nature of the FAERS database, our findings provide further description of the various medications associated with ONJ and elucidate patient demographics associated with the ADR. Additionally, our study identifies cases of several newly described drugs and drug classes that have not been previously described in literature.
Keywords: Adverse drug reaction; Antiangiogenics; Bisphosphonates; Denosumab; Osteonecrosis; m-TOR inhibitors.
© 2023. The Author(s).
Conflict of interest statement
Dr. Eli Ehrenpreis serves as the Chief Executive Officer of E2Bio Life Sciences, a company that is developing Bocaliner, a medical device to improve the treatment of oral mucositis which is a complication of chemotherapy, patent pending. Dr. Ehrenpreis has no additional competing interests to disclose. Dr. Hardeep S. Ahdi, Dr. Thomas Adam Wichelmann and Dr. Sasirekha Pandravada have no competing interests to disclose.
Figures

References
-
- Ganesan K, Bansal P, Goyal A, et al. “Bisphosphonate.” StatPearls. Treasure Island (FL): StatPearls Publishing; updated 2021 Jul 6. Accessed date 26 Sept 2021.
-
- American Association of Oral and Maxillofacial Surgeons. 2014. Medication-Related Osteonecrosis of the Jaw-2014 Update. Retrieved from https://www.aaoms.org/docs/govt_affairs/advocacy_white_papers/mronj_posi.... Accessed date 10/07/2021.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

