Magrolimab in Combination With Azacitidine in Patients With Higher-Risk Myelodysplastic Syndromes: Final Results of a Phase Ib Study
- PMID: 36888930
- PMCID: PMC10414740
- DOI: 10.1200/JCO.22.01794
Magrolimab in Combination With Azacitidine in Patients With Higher-Risk Myelodysplastic Syndromes: Final Results of a Phase Ib Study
Abstract
Purpose: Magrolimab is a monoclonal antibody that blocks cluster of differentiation 47, a don't-eat-me signal overexpressed on cancer cells. Cluster of differentiation 47 blockade by magrolimab promotes macrophage-mediated phagocytosis of tumor cells and is synergistic with azacitidine, which increases expression of eat-me signals. We report final phase Ib data in patients with untreated higher-risk myelodysplastic syndromes (MDS) treated with magrolimab and azacitidine (ClinicalTrials.gov identifier: NCT03248479).
Patients and methods: Patients with previously untreated Revised International Prognostic Scoring System intermediate-/high-/very high-risk MDS received magrolimab intravenously as a priming dose (1 mg/kg) followed by ramp-up to a 30 mg/kg once-weekly or once-every-2-week maintenance dose. Azacitidine 75 mg/m2 was administered intravenously/subcutaneously once daily on days 1-7 of each 28-day cycle. Primary end points were safety/tolerability and complete remission (CR) rate.
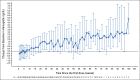
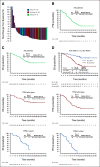
Results: Ninety-five patients were treated. Revised International Prognostic Scoring System risk was intermediate/high/very high in 27%, 52%, and 21%, respectively. Fifty-nine (62%) had poor-risk cytogenetics and 25 (26%) had TP53 mutation. The most common treatment-emergent adverse effects included constipation (68%), thrombocytopenia (55%), and anemia (52%). Median hemoglobin change from baseline to first postdose assessment was -0.7 g/dL (range, -3.1 to +2.4). CR rate and overall response rate were 33% and 75%, respectively. Median time to response, duration of CR, duration of overall response, and progression-free survival were 1.9, 11.1, 9.8, and 11.6 months, respectively. Median overall survival (OS) was not reached with 17.1-month follow-up. In TP53-mutant patients, 40% achieved CR with median OS of 16.3 months. Thirty-four patients (36%) had allogeneic stem-cell transplant with 77% 2-year OS.
Conclusion: Magrolimab + azacitidine was well tolerated with promising efficacy in patients with untreated higher-risk MDS, including those with TP53 mutations. A phase III trial of magrolimab/placebo + azacitidine is ongoing (ClinicalTrials.gov identifier: NCT04313881 [ENHANCE]).
Conflict of interest statement
The following represents disclosure information provided by authors of this manuscript. All relationships are considered compensated unless otherwise noted. Relationships are self-held unless noted. I = Immediate Family Member, Inst = My Institution. Relationships may not relate to the subject matter of this manuscript. For more information about ASCO's conflict of interest policy, please refer to
Open Payments is a public database containing information reported by companies about payments made to US-licensed physicians (
No other potential conflicts of interest were reported.
Figures

References
-
- Aubrey BJ, Brunner AM: SOHO state of the art and next questions: Treatment of higher-risk myelodysplastic syndromes. Clin Lymphoma Myeloma Leuk 22:P869-P877, 2022 - PubMed
-
- Silverman LR, Demakos EP, Peterson BL, et al. : Randomized controlled trial of azacitidine in patients with the myelodysplastic syndrome: A study of the Cancer and Leukemia Group B. J Clin Oncol 20:2429-2440, 2002 - PubMed
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

