An airway-to-brain sensory pathway mediates influenza-induced sickness
- PMID: 36890237
- PMCID: PMC10033449
- DOI: 10.1038/s41586-023-05796-0
An airway-to-brain sensory pathway mediates influenza-induced sickness
Abstract
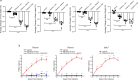
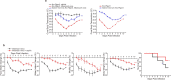
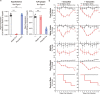
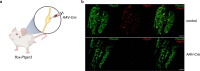
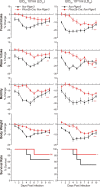
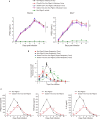
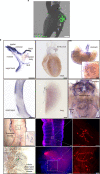
Pathogen infection causes a stereotyped state of sickness that involves neuronally orchestrated behavioural and physiological changes1,2. On infection, immune cells release a 'storm' of cytokines and other mediators, many of which are detected by neurons3,4; yet, the responding neural circuits and neuro-immune interaction mechanisms that evoke sickness behaviour during naturalistic infections remain unclear. Over-the-counter medications such as aspirin and ibuprofen are widely used to alleviate sickness and act by blocking prostaglandin E2 (PGE2) synthesis5. A leading model is that PGE2 crosses the blood-brain barrier and directly engages hypothalamic neurons2. Here, using genetic tools that broadly cover a peripheral sensory neuron atlas, we instead identified a small population of PGE2-detecting glossopharyngeal sensory neurons (petrosal GABRA1 neurons) that are essential for influenza-induced sickness behaviour in mice. Ablating petrosal GABRA1 neurons or targeted knockout of PGE2 receptor 3 (EP3) in these neurons eliminates influenza-induced decreases in food intake, water intake and mobility during early-stage infection and improves survival. Genetically guided anatomical mapping revealed that petrosal GABRA1 neurons project to mucosal regions of the nasopharynx with increased expression of cyclooxygenase-2 after infection, and also display a specific axonal targeting pattern in the brainstem. Together, these findings reveal a primary airway-to-brain sensory pathway that detects locally produced prostaglandins and mediates systemic sickness responses to respiratory virus infection.
© 2023. The Author(s).
Conflict of interest statement
S.D.L. is a consultant for Kallyope, Inc. The other authors declare no competing interests.
Figures




Comment in
-
How the brain senses a flu infection - and orders the body to rest.Nature. 2023 Mar;615(7952):385. doi: 10.1038/d41586-023-00675-0. Nature. 2023. PMID: 36890322 No abstract available.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

