Serotonin and the ventilatory effects of etonogestrel, a gonane progestin, in a murine model of congenital central hypoventilation syndrome
- PMID: 36896185
- PMCID: PMC9989262
- DOI: 10.3389/fendo.2023.1077798
Serotonin and the ventilatory effects of etonogestrel, a gonane progestin, in a murine model of congenital central hypoventilation syndrome
Abstract
Introduction: Congenital Central Hypoventilation Syndrome, a rare disease caused by PHOX2B mutation, is associated with absent or blunted CO2/H+ chemosensitivity due to the dysfunction of PHOX2B neurons of the retrotrapezoid nucleus. No pharmacological treatment is available. Clinical observations have reported non-systematic CO2/H+ chemosensitivity recovery under desogestrel.
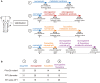
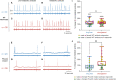
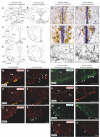
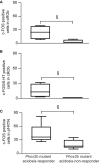
Methods: Here, we used a preclinical model of Congenital Central Hypoventilation Syndrome, the retrotrapezoid nucleus conditional Phox2b mutant mouse, to investigate whether etonogestrel, the active metabolite of desogestrel, led to a restoration of chemosensitivity by acting on serotonin neurons known to be sensitive to etonogestrel, or retrotrapezoid nucleus PHOX2B residual cells that persist despite the mutation. The influence of etonogestrel on respiratory variables under hypercapnia was investigated using whole-body plethysmographic recording. The effect of etonogestrel, alone or combined with serotonin drugs, on the respiratory rhythm of medullary-spinal cord preparations from Phox2b mutants and wildtype mice was analyzed under metabolic acidosis. c-FOS, serotonin and PHOX2B were immunodetected. Serotonin metabolic pathways were characterized in the medulla oblongata by ultra-high-performance liquid chromatography.
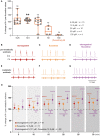
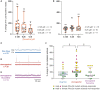
Results: We observed etonogestrel restored chemosensitivity in Phox2b mutants in a non-systematic way. Histological differences between Phox2b mutants with restored chemosensitivity and Phox2b mutant without restored chemosensitivity indicated greater activation of serotonin neurons of the raphe obscurus nucleus but no effect on retrotrapezoid nucleus PHOX2B residual cells. Finally, the increase in serotonergic signaling by the fluoxetine application modulated the respiratory effect of etonogestrel differently between Phox2b mutant mice and their WT littermates or WT OF1 mice, a result which parallels with differences in the functional state of serotonergic metabolic pathways between these different mice.
Discussion: Our work thus highlights that serotonin systems were critically important for the occurrence of an etonogestrel-restoration, an element to consider in potential therapeutic intervention in Congenital Central Hypoventilation Syndrome patients.
Keywords: CO2/H+ chemosensitivity; central breathing disorder; mouse model; progestin; serotoninergic systems.
Copyright © 2023 Casciato, Bianchi, Reverdy, Joubert, Delucenay-Clarke, Parrot, Ramanantsoa, Sizun, Matrot, Straus, Similowski, Cayetanot and Bodineau.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures


References
-
- Dubreuil V, Ramanantsoa N, Trochet D, Vaubourg V, Amiel J, Gallego J, et al. . A human mutation in Phox2b causes lack of CO2 chemosensitivity, fatal central apnea, and specific loss of parafacial neurons. Proc Natl Acad Sci U.S.A. (2008) 105:1067–72. doi: 10.1073/pnas.0709115105 - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Supplementary concepts
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

