Immune correlates analysis of a phase 3 trial of the AZD1222 (ChAdOx1 nCoV-19) vaccine
- PMID: 36899062
- PMCID: PMC10005913
- DOI: 10.1038/s41541-023-00630-0
Immune correlates analysis of a phase 3 trial of the AZD1222 (ChAdOx1 nCoV-19) vaccine
Abstract
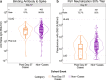
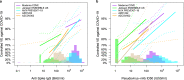
In the phase 3 trial of the AZD1222 (ChAdOx1 nCoV-19) vaccine conducted in the U.S., Chile, and Peru, anti-spike binding IgG concentration (spike IgG) and pseudovirus 50% neutralizing antibody titer (nAb ID50) measured four weeks after two doses were assessed as correlates of risk and protection against PCR-confirmed symptomatic SARS-CoV-2 infection (COVID-19). These analyses of SARS-CoV-2 negative participants were based on case-cohort sampling of vaccine recipients (33 COVID-19 cases by 4 months post dose two, 463 non-cases). The adjusted hazard ratio of COVID-19 was 0.32 (95% CI: 0.14, 0.76) per 10-fold increase in spike IgG concentration and 0.28 (0.10, 0.77) per 10-fold increase in nAb ID50 titer. At nAb ID50 below the limit of detection (< 2.612 IU50/ml), 10, 100, and 270 IU50/ml, vaccine efficacy was -5.8% (-651%, 75.6%), 64.9% (56.4%, 86.9%), 90.0% (55.8%, 97.6%) and 94.2% (69.4%, 99.1%). These findings provide further evidence towards defining an immune marker correlate of protection to help guide regulatory/approval decisions for COVID-19 vaccines.
© 2023. The Author(s).
Conflict of interest statement
E.J.K., I.H., S.S., A.M.S., J.M., and T.V. are employees of AstraZeneca and hold/may hold stock in AstraZeneca.
Figures




References
Grants and funding
- UM1 AI148450/AI/NIAID NIH HHS/United States
- UM1 AI148372/AI/NIAID NIH HHS/United States
- C0000008/CL/CLC NIH HHS/United States
- UM1 AI069470/AI/NIAID NIH HHS/United States
- UM1 AI068614/AI/NIAID NIH HHS/United States
- UM1 AI068618/AI/NIAID NIH HHS/United States
- R37 AI054165/AI/NIAID NIH HHS/United States
- UM1 AI148684/AI/NIAID NIH HHS/United States
- UM1 AI068635/AI/NIAID NIH HHS/United States
- UM1 AI068619/AI/NIAID NIH HHS/United States
- S10 OD028685/OD/NIH HHS/United States
- U01 AI069470/AI/NIAID NIH HHS/United States
- UM1 AI068636/AI/NIAID NIH HHS/United States
- UM1 AI148574/AI/NIAID NIH HHS/United States
LinkOut - more resources
Full Text Sources
Miscellaneous

