Healthcare Resource Utilization in Patients with Newly Diagnosed Atrial Fibrillation: A Global Analysis from the GARFIELD-AF Registry
- PMID: 36900643
- PMCID: PMC10000823
- DOI: 10.3390/healthcare11050638
Healthcare Resource Utilization in Patients with Newly Diagnosed Atrial Fibrillation: A Global Analysis from the GARFIELD-AF Registry
Abstract
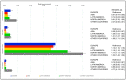
The management of atrial fibrillation (AF), the most common sustained arrhythmia, impacts healthcare resource utilization (HCRU). This study aims to estimate global resource use in AF patients, using the GARFIELD-AF registry. A prospective cohort study was conducted to characterize HCRU in AF patients enrolled in sequential cohorts from 2012 to 2016 in 35 countries. Components of HCRU studied were hospital admissions, outpatient care visits, and diagnostic and interventional procedures occurring during follow-up. AF-related HCRU was reported as the percentage of patients demonstrating at least one event and was quantified as rate-per-patient-per-year (PPPY) over time. A total of 49,574 patients was analyzed, having an overall median follow-up of 719 days. Almost all patients (99.5%) had at least one outpatient care visit, while hospital admissions were the second most frequent medical contact, with similar proportions in North America (37.5%) and Europe (37.2%), and slightly higher in the other GARFIELD-AF countries (42.0%; namely Australia, Egypt, and South Africa). Asia and Latin America showed lower percentages of hospitalizations, outpatient care visits, and diagnostic and interventional procedures. Analyses of GARFIELD-AF highlighted the vast AF-related HCRU, underlying significant geographical differences in the type, quantity, and frequency of AF-related HCRU. These differences were likely attributable to health service availability and differing models of care.
Keywords: atrial fibrillation; healthcare resource utilization; inpatient care; outpatient care.
Conflict of interest statement
L.G.M. received grants and personal fees from Bayer AG, Daiichi Sankyo, Pfizer and Boehringer Ingelheim. A.J.C. received institutional grants and personal fees from Bayer, Boehringer Ingelheim, Pfizer/BMS, Daiichi Sankyo; F.W.A.V. received grants from Bayer Healthcare; personal fees from Bayer Healthcare, BMS/Pfizer, DaiichiSankyo, and Boehringer-Ingelheim; J.P.B. received personal fees from the Thrombosis Research Institute; A.G.G.T. received personal fees from Bayer Healthcare, Janssen Pharmaceutical Research & Development, and Portola; W.H. personal fees from Bayer AG; S.Z.G. received research support from Boehringer-Ingelheim, BMS, BTG EKOS, Daiichi, Janssen, NHLBI, and the Thrombosis Research Institute; S.Z.G. also served as a consultant for Agile, Bayer, Boehringer-Ingelheim, BMS, Daiichi, Janssen and Zafgen; S.G. received personal fees from Thrombosis Research Institute and the American Heart Association, as well as grants from Sanofi, Pfizer, Ono, Bristol Myer Squibb, the Vehicle Racing Commemorative Foundation and Nakatani Foundation for Advancement of Measuring Technologies in Biomedical Engineering; K.S.P. serves as consultant for Thrombosis Research Institute, AstraZeneca, and Bayer; B.J.G. is a data safety monitoring board member at Mount Sinai St Luke’s, Boston Scientific Corporation, St Jude Medical Inc., Janssen Research & Development LLC, Thrombosis Research Institute, Duke Clinical Research Institute, Duke University, Kowa Research Institute Inc, Cardiovascular Research Foundation, and Medtronic; he also received general consulting for Janssen Scientific Affairs, Xenon Pharmaceuticals and Sirtex Medical Limited; K.A.A.F. has received grants and personal fees from Bayer/Janssen and AstraZeneca and personal fees from Sanofi/Regeneron and Verseon; S.H. received personal fees from Aspen, Bayer, BMS, Daiichi-Sankyo, Portola, Sanofi; M.v.E. is an employee of Bayer AG; A.K.K. received research support from Bayer AG and Sanofi, and personal fees from Bayer AG, Janssen, Pfizer, Sanofi, Verseon and Anthos Therapeutics; P.C., P.F., S.V. and G.K. declare no conflicts of interest.
Figures

References
-
- Roth G.A., Mensah G.A., Johnson C.O., Addolorato G., Ammirati E., Baddour L.M., Barengo N.C., Beaton A.Z., Benjamin E.Z., Benzinger C.P., et al. Global Burden of Cardiovascular Diseases and Risk Factors, 1990 to 2019: Update from the Global Burden of Disease 2019 Study. JACC J. Am. Coll. Cardiol. 2020;76:2982–3021. doi: 10.1016/j.jacc.2020.11.010. - DOI - PMC - PubMed
-
- Li H., Song X., Liang Y., Bai X., Liu-Huo W.S., Tang C., Chen W., Zhao L. Global, regional, and national burden of disease study of atrial fibrillation/flutter, 1990-2019: Results from a global burden of disease study, 2019. BMC Public Health. 2022;22:2015. doi: 10.1186/s12889-022-14403-2. - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources

