Histone Deacetylases: Molecular Mechanisms and Therapeutic Implications for Muscular Dystrophies
- PMID: 36901738
- PMCID: PMC10002075
- DOI: 10.3390/ijms24054306
Histone Deacetylases: Molecular Mechanisms and Therapeutic Implications for Muscular Dystrophies
Abstract
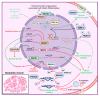
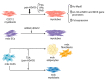
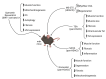
Histone deacetylases (HDACs) are enzymes that regulate the deacetylation of numerous histone and non-histone proteins, thereby affecting a wide range of cellular processes. Deregulation of HDAC expression or activity is often associated with several pathologies, suggesting potential for targeting these enzymes for therapeutic purposes. For example, HDAC expression and activity are higher in dystrophic skeletal muscles. General pharmacological blockade of HDACs, by means of pan-HDAC inhibitors (HDACi), ameliorates both muscle histological abnormalities and function in preclinical studies. A phase II clinical trial of the pan-HDACi givinostat revealed partial histological improvement and functional recovery of Duchenne Muscular Dystrophy (DMD) muscles; results of an ongoing phase III clinical trial that is assessing the long-term safety and efficacy of givinostat in DMD patients are pending. Here we review the current knowledge about the HDAC functions in distinct cell types in skeletal muscle, identified by genetic and -omic approaches. We describe the signaling events that are affected by HDACs and contribute to muscular dystrophy pathogenesis by altering muscle regeneration and/or repair processes. Reviewing recent insights into HDAC cellular functions in dystrophic muscles provides new perspectives for the development of more effective therapeutic approaches based on drugs that target these critical enzymes.
Keywords: Duchenne Muscular Dystrophy; clinical trials; histone deacetylase; muscular dystrophies.
Conflict of interest statement
T.A.M. is on the scientific advisory boards of Artemes Bio and Eikonizo Therapeutics, received funding from Italfarmaco for an unrelated project, and has a subcontract from Eikonizo Therapeutics related to an SBIR grant from the National Institutes of Health (HL154959).
Figures
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

