Effect of SARS-CoV-2 mRNA-Vaccine on the Induction of Myocarditis in Different Murine Animal Models
- PMID: 36902442
- PMCID: PMC10002951
- DOI: 10.3390/ijms24055011
Effect of SARS-CoV-2 mRNA-Vaccine on the Induction of Myocarditis in Different Murine Animal Models
Abstract
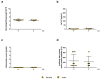
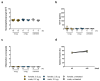
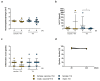
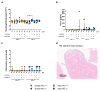
In the course of the SARS-CoV-2 pandemic, vaccination safety and risk factors of SARS-CoV-2 mRNA-vaccines were under consideration after case reports of vaccine-related side effects, such as myocarditis, which were mostly described in young men. However, there is almost no data on the risk and safety of vaccination, especially in patients who are already diagnosed with acute/chronic (autoimmune) myocarditis from other causes, such as viral infections, or as a side effect of medication and treatment. Thus, the risk and safety of these vaccines, in combination with other therapies that could induce myocarditis (e.g., immune checkpoint inhibitor (ICI) therapy), are still poorly assessable. Therefore, vaccine safety, with respect to worsening myocardial inflammation and myocardial function, was studied in an animal model of experimentally induced autoimmune myocarditis. Furthermore, it is known that ICI treatment (e.g., antibodies (abs) against PD-1, PD-L1, and CTLA-4, or a combination of those) plays an important role in the treatment of oncological patients. However, it is also known that treatment with ICIs can induce severe, life-threatening myocarditis in some patients. Genetically different A/J (most susceptible strain) and C57BL/6 (resistant strain) mice, with diverse susceptibilities for induction of experimental autoimmune myocarditis (EAM) at various age and gender, were vaccinated twice with SARS-CoV-2 mRNA-vaccine. In an additional A/J group, an autoimmune myocarditis was induced. In regard to ICIs, we tested the safety of SARS-CoV-2 vaccination in PD-1-/- mice alone, and in combination with CTLA-4 abs. Our results showed no adverse effects related to inflammation and heart function after mRNA-vaccination, independent of age, gender, and in different mouse strains susceptible for induction of experimental myocarditis. Moreover, there was no worsening effect on inflammation and cardiac function when EAM in susceptible mice was induced. However, in the experiments with vaccination and ICI treatment, we observed, in some mice, low elevation of cardiac troponins in sera, and low scores of myocardial inflammation. In sum, mRNA-vaccines are safe in a model of experimentally induced autoimmune myocarditis, but patients undergoing ICI therapy should be closely monitored when vaccinated.
Keywords: SARS-CoV-2; mRNA vaccine; myocardial function; myocarditis; side effects.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


References
-
- Ozma M.A., Maroufi P., Khodadadi E., Kose S., Esposito I., Ganbarov K., Dao S., Esposito S., Dal T., Zeinalzadeh E., et al. Clinical manifestation, diagnosis, prevention and control of SARS-CoV-2 (COVID-19) during the outbreak period. Infez. Med. 2020;28:153–165. - PubMed
-
- Poletti P., Tirani M., Cereda D., Trentini F., Guzzetta G., Marziano V., Buoro S., Riboli S., Crottogini L., Piccarreta R., et al. Age-specific SARS-CoV-2 infection fatality ratio and associated risk factors, Italy, February to April 2020. Eurosurveillance. 2020;25:2001383. doi: 10.2807/1560-7917.ES.2020.25.31.2001383. - DOI - PMC - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

