A Review on Porosity Formation in Aluminum-Based Alloys
- PMID: 36903162
- PMCID: PMC10004325
- DOI: 10.3390/ma16052047
A Review on Porosity Formation in Aluminum-Based Alloys
Abstract
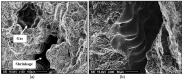
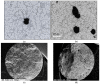
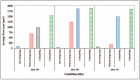
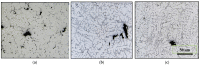
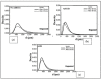
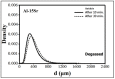
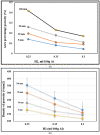
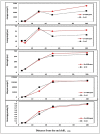
The main objective of this review is to analyze the equations proposed for expressing the effect of various parameters on porosity formation in aluminum-based alloys. These parameters include alloying elements, solidification rate, grain refining, modification, hydrogen content, as well as the applied pressure on porosity formation in such alloys. They are used to establish as precisely as possible a statistical model to describe the resulting porosity characteristics such as the percentage porosity and pore characteristics, as controlled by the chemical composition of the alloy, modification, grain refining, and the casting conditions. The measured parameters of percentage porosity, maximum pore area, average pore area, maximum pore length, and average pore length, which were obtained from statistical analysis, are discussed, and they are supported using optical micrographs, electron microscopic images of fractured tensile bars, as well as radiography. In addition, an analysis of the statistical data is presented. It should be noted that all alloys described were well degassed and filtered prior to casting.
Keywords: grain refining; hydrogen; modification; porosity; solidification rate; statistical modeling.
Conflict of interest statement
The authors declare no conflict of interest.
Figures









References
-
- Liu L., Samuel A.M., Samuel F.H., Doty H.W., Valtierra S. Influence of oxides on porosity formation in Sr-treated Al-Si casting alloys. J. Mater. Sci. 2003;38:1255–1267. doi: 10.1023/A:1022870006721. - DOI
-
- Dinnis C.M., Dahle A.K., Taylor J.A., Otte M.O. The influence of strontium on porosity formation in Al-Si alloys. Metall. Mater. Trans. A. 2004;35:3531–3541. doi: 10.1007/s11661-004-0190-9. - DOI
-
- Shafyei A., Anijdan S.H.M., Bahrami A. Prediction of porosity percent in Al–Si casting alloys using ANN. Mater. Sci. Eng. A. 2006;431:206–210. doi: 10.1016/j.msea.2006.05.150. - DOI
-
- Ye H.I. An overview of the development of Al-Si-Alloy based material for engine applications. J. Mater. Eng. Perform. 2003;12:288–297. doi: 10.1361/105994903770343132. - DOI
-
- Anyalebechi P.N. Hydrogen-induced gas porosity formation in Al–4.5 wt% Cu–1.4 wt% Mg alloy. J. Mater. Sci. 2013;48:5342–5353. doi: 10.1007/s10853-013-7329-2. - DOI
Publication types
LinkOut - more resources
Full Text Sources

