Hydrogen Bond Assisted Three-Component Tandem Reactions to Access N-Alkyl-4-Quinolones
- PMID: 36903552
- PMCID: PMC10005641
- DOI: 10.3390/molecules28052304
Hydrogen Bond Assisted Three-Component Tandem Reactions to Access N-Alkyl-4-Quinolones
Abstract
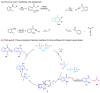
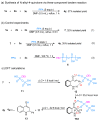
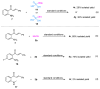
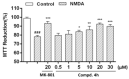
Hydrogen-bonding catalytic reactions have gained great interest. Herein, a hydrogen-bond-assisted three-component tandem reaction for the efficient synthesis of N-alkyl-4-quinolones is described. This novel strategy features the first proof of polyphosphate ester (PPE) as a dual hydrogen-bonding catalyst and the use of readily available starting materials for the preparation of N-alkyl-4-quinolones. The method provides a diversity of N-alkyl-4-quinolones in moderate to good yields. The compound 4h demonstrated good neuroprotective activity against N-methyl-ᴅ-aspartate (NMDA)-induced excitotoxicity in PC12 cells.
Keywords: N-alkyl-4-quinolones; hydrogen bond; neuroprotective activity; polyphosphate ester; polyphosphoric acid.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

References
-
- Koyama J., Toyokuni I., Tagahara K. Synthesis of 2-arylquinoline and 2-aryl-4-quinolone alkaloids via Diels-Alder reaction of 1,2,3-benzotriazine with enamines. Chem. Pharm. Bull. 1999;47:1029–1038. doi: 10.1248/cpb.47.1038. - DOI
MeSH terms
Substances
Grants and funding
- 32160104/National Natural Science Foundation of China
- QKHZC[2021] general 424/Project of Guizhou Science and Technology Platform and Talent Team
- QKHZC[2021] general 129/Project of Guizhou Science and Technology Platform and Talent Team
- QKHJC[2021] general 512/Guizhou Provincial Natural Science Foundation
- QJHKYZ[2020]018/Project of Key Laboratory for Kharacteristics of Colleges and Universities of Guizhou Provincial Department of Education
LinkOut - more resources
Full Text Sources

