This is a preprint.
Mode-based morphometry: A multiscale approach to mapping human neuroanatomy
- PMID: 36909539
- PMCID: PMC10002616
- DOI: 10.1101/2023.02.26.529328
Mode-based morphometry: A multiscale approach to mapping human neuroanatomy
Update in
-
Mode-based morphometry: A multiscale approach to mapping human neuroanatomy.Hum Brain Mapp. 2024 Mar;45(4):e26640. doi: 10.1002/hbm.26640. Hum Brain Mapp. 2024. PMID: 38445545 Free PMC article.
Abstract
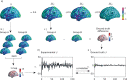
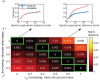
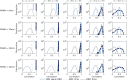
Voxel-based morphometry (VBM) and surface-based morphometry (SBM) are two widely used neuroimaging techniques for investigating brain anatomy. These techniques rely on statistical inferences at individual points (voxels or vertices), clusters of points, or a priori regions-of-interest. They are powerful tools for describing brain anatomy, but offer little insights into the generative processes that shape a particular set of findings. Moreover, they are restricted to a single spatial resolution scale, precluding the opportunity to distinguish anatomical variations that are expressed across multiple scales. Drawing on concepts from classical physics, here we develop an approach, called mode-based morphometry (MBM), that can describe any empirical map of anatomical variations in terms of the fundamental, resonant modes--eigenmodes--of brain anatomy, each tied to a specific spatial scale. Hence, MBM naturally yields a multiscale characterization of the empirical map, affording new opportunities for investigating the spatial frequency content of neuroanatomical variability. Using simulated and empirical data, we show that the validity and reliability of MBM are either comparable or superior to classical vertex-based SBM for capturing differences in cortical thickness maps between two experimental groups. Our approach thus offers a robust, accurate, and informative method for characterizing empirical maps of neuroanatomical variability that can be directly linked to a generative physical process.
Figures








References
-
- Ashburner J., Friston K. J., Voxel-based morphometry–the methods, NeuroImage 11 (2000) 805–821. - PubMed
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
